ENGLISH-MSN-1-FLUID AND ELECTROLYATE-UPLOAD
Fluid and Electrolyte Balance and Imbalance:
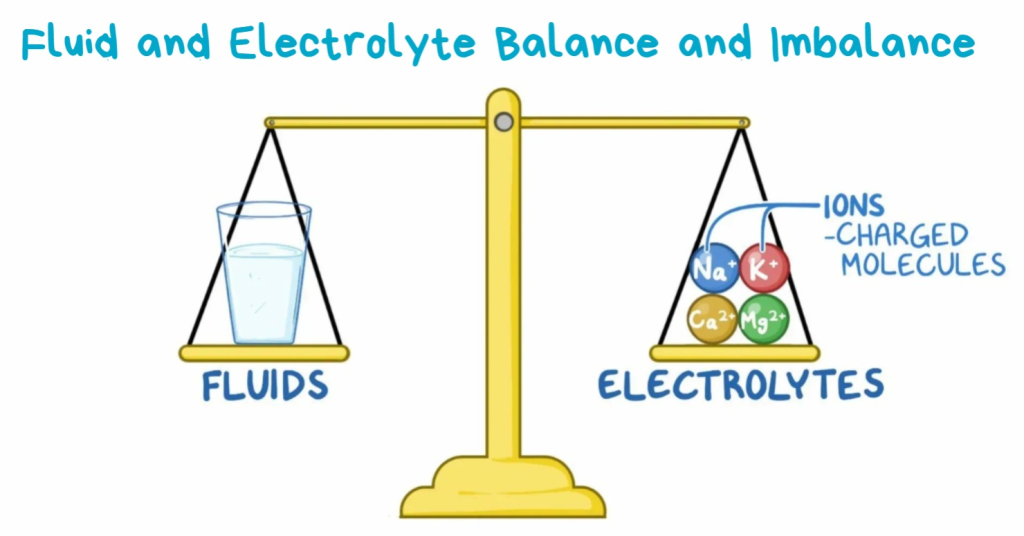
a) Water contents of body, electrolyte and Acid – Base balance
b) Homeostasis
c) Review mechanism of regulating fluid and electrolyte movement
d) Fluid and electrolyte Acid-Base imbalance and its management
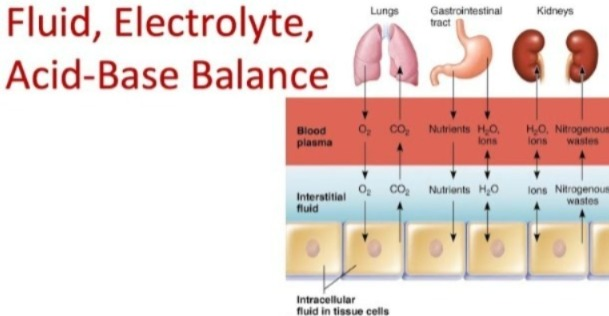
INTRODUCTION OF FLUID ELECTROLYTE AND ACID BASE BALANCE balance):
Definition:
1) Osmolarity:
One liter solution The number of solute present in a solution is called osmolarity.
OR
Osmolarity is a measure of the total number of solute particles in a solution that are dissolved in one liter of solution. That is, Osmolarity indicates the concentration of a solution, which is the number of osmotically active particles present per liter of solution after the solute substances are dissolved in a solvent such as water. Osmolarity is measured in milliosmoles per liter of solution (mOsm/L). It is important for understanding the fluid balance between solutes and water in body fluids and the movement of water across cell membranes.
2) Osmolality:

Osmolality refers to the concentration of total solute particles present in a fluid and is measured in milliosmoles per kilogram (mOsm/kg) of solute per kilogram of solvent. Osmolality plays an important role in maintaining osmotic pressure in the body, controlling fluid balance, and evaluating different organ functions such as kidney function and hydration status. This parameter is determined based on the amount of solutes such as sodium, glucose, and urea and is used in clinical practice to analyze biological fluids such as blood and urine.
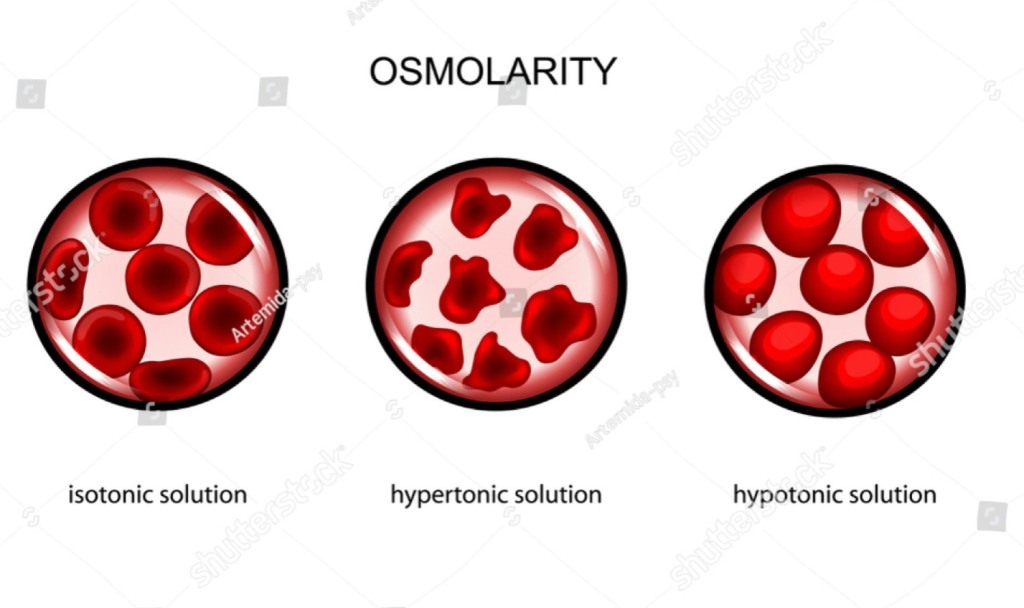
3) Isotonic:
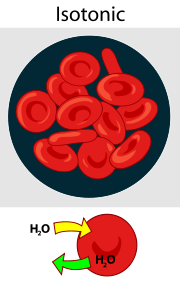
Isotonic means a solution whose Osmotic Pressure (Osmotic Pressure) or Solute Concentration is the same as the Intracellular Fluid or Body Fluid of the body. In other words, an Isotonic solution is a solution in which water molecules can neither enter nor leave the cell because the concentration of solute particles on both sides of the cell membrane is equal. Therefore, the shape and volume of the cell remain stable. For example, 0.9% Sodium Chloride solution is considered an Isotonic solution.
OR
If the osmolarity of a solution is the same as the osmolarity of body fluid (300 mol/liter), then it is called an isotonic fluid.
( isotonic solutions:=osmolarity is equal to 300 mol/liter)
Ex. 0.9 % NaCl, Ringer Lactate (RL), Dextrose 5 percent
4)hypotonic:
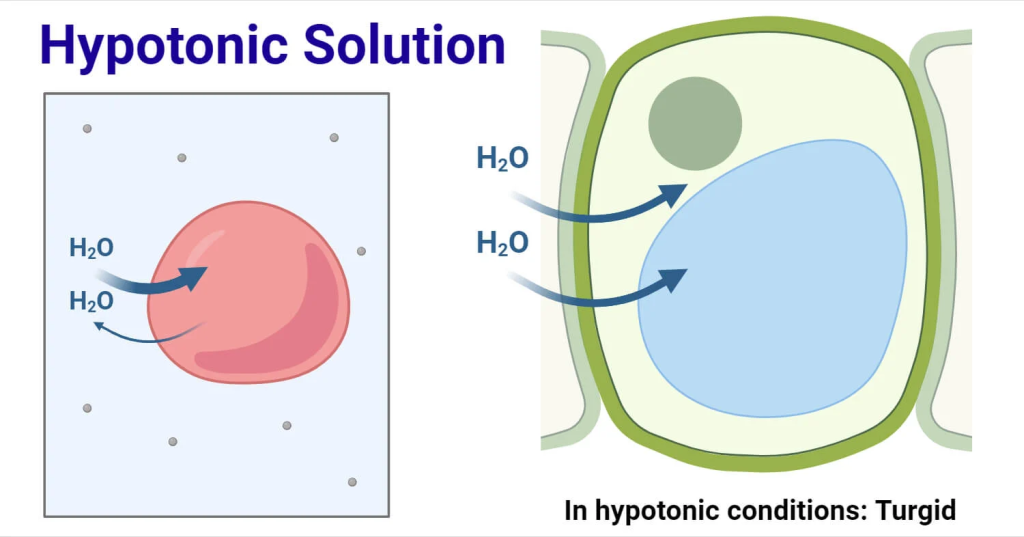
Hypotonic is a medical term used for a condition or solution in which the concentration of solutes inside the solution is low compared to the solutes inside the cell. As a result, when a hypotonic solution comes into contact with a cell, water enters the cell through the process of osmosis, causing the cell to swell and sometimes even burst (lysis). Hypotonic solutions are used in various medical treatments, especially for hydration, but they must be used with caution because excessive use can damage cells.
OR
A fluid is called a hypotonic solution if its osmolarity is less than that of body fluid.
(hypotonic solutions: osmolarity is less than 300 mol/liter)
A hypotonic solution is one that causes the cell to swell.
0.45% NaCl,0.33%, NaCl,0.2 % NaCl,2.5 %Dextrose
5) hypertonic solutions (hypertonic):
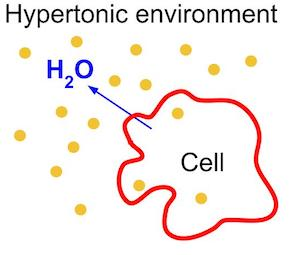
Hypertonic is a medical term used for a solution in which the concentration of solutes inside the solution is higher compared to the solutes inside the cell. As a result, when a hypertonic solution comes into contact with a cell, water moves out of the cell by a process called osmosis, causing cell shrinkage or crenation. Hypertonic solutions are used in some medical conditions, such as to reduce intracranial pressure, but they should be used with caution under the guidance of a specialist because excessive use can dehydrate cells.
OR
If the osmolarity of a fluid is greater than the osmolarity of the body fluid, it is called a hypertonic solution.
( hypertonic solutions: osmolarity is greater than 300 mol/ liter).
This means that this solution shrinks the cell.
3% NaCl,5% NaCl, Mannitol
Fluid, electrolyte and acid-base balance is a dynamic process and is crucial in life.
Assessing the patient’s fluid and electrolyte status And maintaining it is a major responsibility of nursing.
If the balance of fluid and electrolytes in the body is normal, then the function of the body will remain normal.
But when there is even a slight alteration in fluid and electrolytes, the normal physiology of the body also gets altered.
If the body fluid intake and fluid output are normal, then there is a balance between both.
For the promotion of health, It is very important to maintain the amount of fluid and acid-base balance.
Continuous dynamic changes occur in the body.
BODY FLUID:
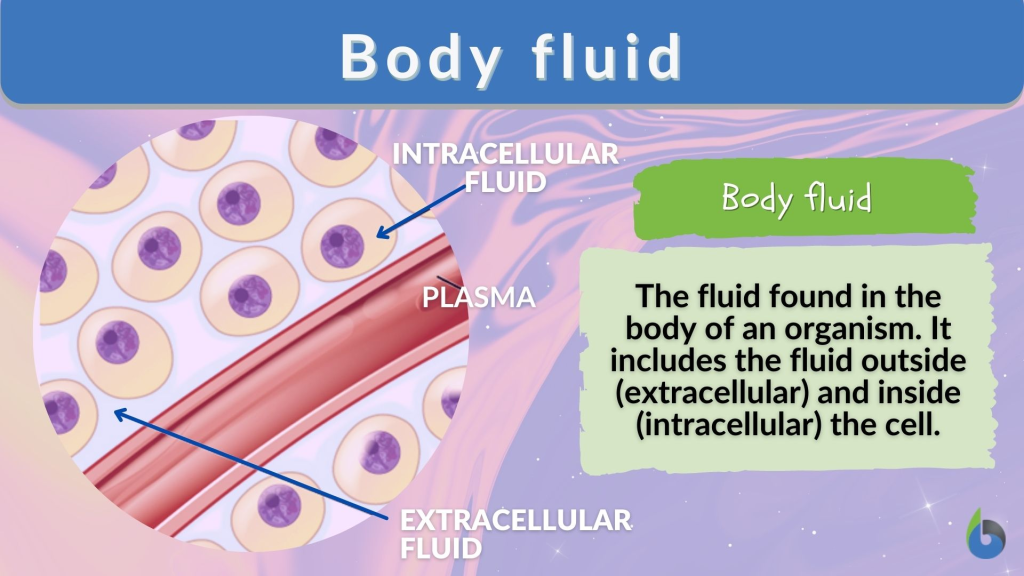
- A male’s body contains 60% water.
- While a female’s body contains 50% water.
- 75% of it is in the brain,
70 to 75% of it is in the muscles,
and 22% is in the bones. - 50% in elderly people less water than and an infant has 70% to 80% water.
1) Extracellular fluid (Fluid outside the cell) :
35-40% is (14 liter).
•Interstitial fluid :10.5liter, (situated between two cells)
•plasma: 3liter ,(plasma)
transcellular fluid: 0.5liter. (cerebrospinal fluid. synovial fluid (joint fluid) aqueous humor (ocular fluid)
2) Intracellular fluid:
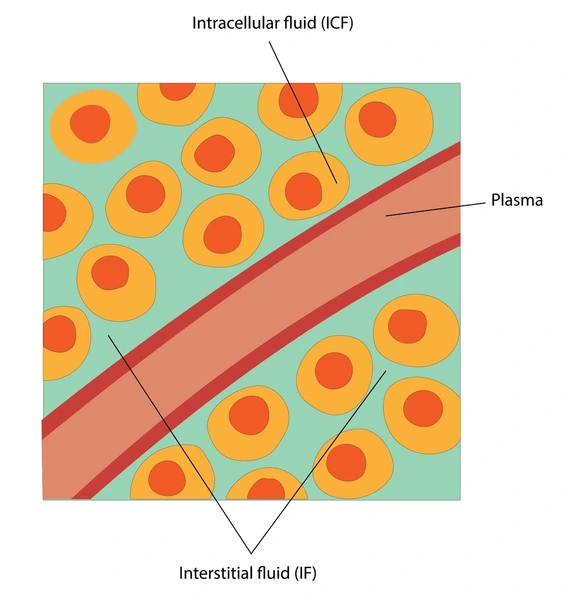
60-65% (28 liters)
The body contains 6-10 liters of lymph and 3.5-5 liters of blood.

Explain the fluid balance :-
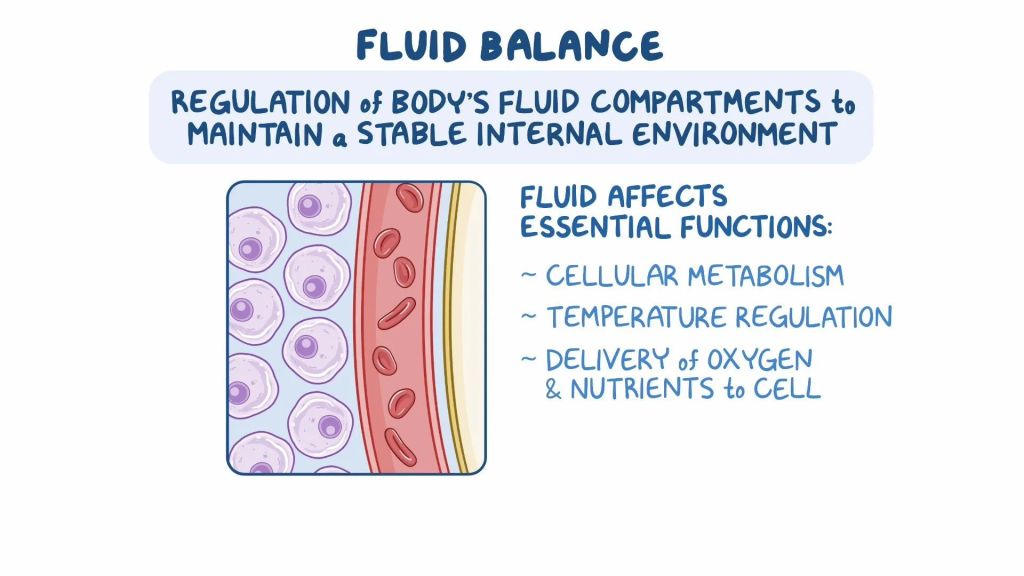
- The body’s fluid is constantly lost and the fluid balance is normalized by whatever liquids and food the human being ingests.
- Fluid balance is the balance in which water is ingested into the body.
Water is excreted from the body through any drink, food, etc., through urine and other waste products, thus maintaining the fluid balance in the body. - Fluid is mainly excreted from the kidneys, lungs and skin. is.
- Body heat is removed from the skin and lungs through the process of evaporation of water
- Urea and other metabolic waste products are excreted (removed) by the kidneys.
- Body fluids play an important role in supplying oxygen and nutrients to the cells.
- Bodily fluids include aqueous humor (aqueous humor: located in the anterior chamber of the eye),
- Vitrous humor ( Vitreous humor : It is located in the posterior chamber of the eye),
- Bile ( Bile),
- blood serum ( Blood serum),
- Breast milk ( Breast milk),
- Cerebrospinal fluid ( Cerebro spinal fluid),
- Cerumen ( earwax),
- Endolymph and perilymph,
- Gastric juice (gastric juice),
- Mucous (mucus),
- Peritoneal fluid (fluid around the abdomen),
- Pleural fluid (fluid around the lungs),
- Saliva ( saliva),
- Sebum ( skin oil),
- Semen ( semen),
- Sweat ( sweat),
- Tears ( tears),
- Vaginal secretion ( vaginal secretion),
- Vomit, urine , etc Located in the body and maintains homeostasis of the body’s internal environment.
Body fluid has been divided into Two compartments:
- 1) intracellular fluid (ICF: fluid inside the cell),
- 2) extracellular fluid (ECF: fluid outside the cell)
1) Intracellular fluid:
- Intracellular fluid is located inside the cell.
- The cell membrane and cellular metabolism control the cellular constituents.
- Intracellular fluid is a two-by-three ( 2/3).
- Intracellular fluid contains potassium, magnesium, phosphate, organic anions, and proteins etc.
- If the body is 60% water, then intracellular fluid is 40% of the body weight.
- Intracellular fluid is a mixture of each cell but it is the same in all cells.
- by volume intracellular Fluid is the largest body fluid compartment of the body.
2) Extracellular fluid:
- The fluid outside the cells is called extracellular fluid.
- Extracellular fluid accounts for 1/3 of body water.
- Extracellular fluid accounts for 20% of body weight.
- Extracellular fluid contains sodium, chloride, bicarbonate, and other substances such as oxygen, glucose, fatty acids, and amino acids. is.
Extracellular fluid is further divided into three parts.
- 1) interstitial fluid( ISF),
- 2) Intravascular fluid (plasma),
- 3) transcellular fluid.
1)Interstitial fluid (interstitial fluid):
- Interstitial fluid is located around the cell.
- This interstitial fluid (interstitial fluid) is located 3/4 of the extracellular fluid (extra cellular fluid).
2)Intravascular fluid (plasma):
- Fluid and electrolytes move between the interstitial and intravascular fluids and are called plasma.
- Plasma circulates as the extracellular component of blood.
- And it is 1/4 of the extracellular fluid.
3)transcellular fluid (transcellular Fluid:
- This is the set of fluids that are outside the normal compartment.
- And this is about one to two liters (1-2 liters).
Which includes cerebrospinal fluid (CSF : CEREBRO SPINAL FLUID), digestive juice (DIAGESTIVE JUICE), MUCOUS.
Explain the function of body fluid:
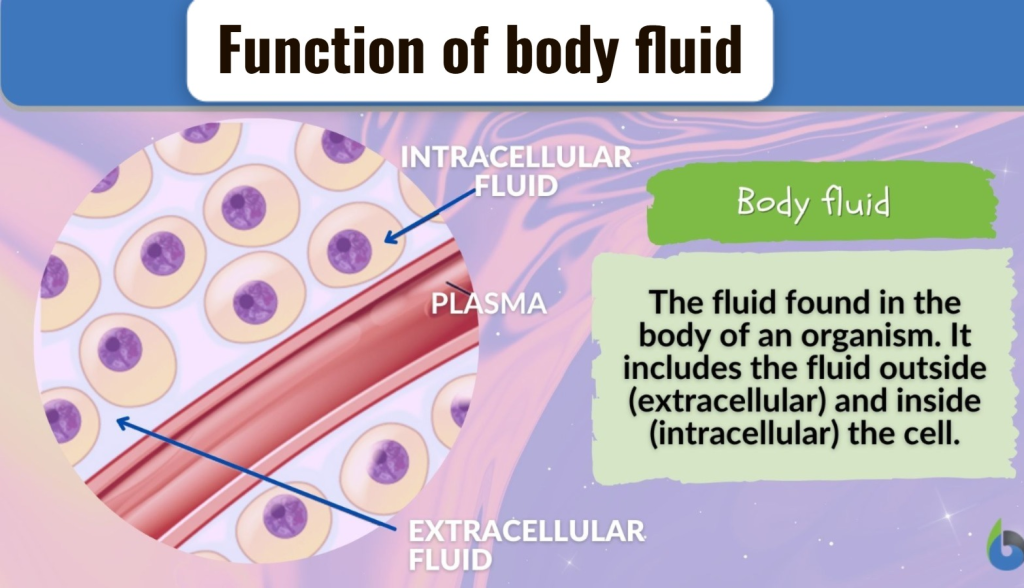
1) Blood carries nutrients to the cells and removes waste products from the body.
2) Body fluids maintain normal blood volume and blood pressure and maintain the balance between them.
3) Body fluids maintain body temperature.
4) This body fluid evaporates fluid through sweating and maintains the body’s homeostasis.
5) Body fluid works as an aqueous medium for cells.
6) It works as a solvent for many chemical reactions.
7) It helps in the digestion of food through fluid.
8) Fluid works for the excretion of waste products.
9) Fluid acts as a cushion for vital organs and protects them.
10) Fluid helps convert food into energy.
11) Fluid acts as a cushion between the joints of the bones.
12) Fluid works to eliminate waste products.
13) Fluid improves the health of the intestine and due to this, constipation is eliminated.
14) Fluid flushes the kidneys due to which the waste products of metabolism are removed from the body.
15) Fluid works to keep the body’s vital functions normal.
