BSC SEM 4 UNIT 2 PATHOLOGY 2 & GENETICS.
UNIT 2 Clinical Pathology
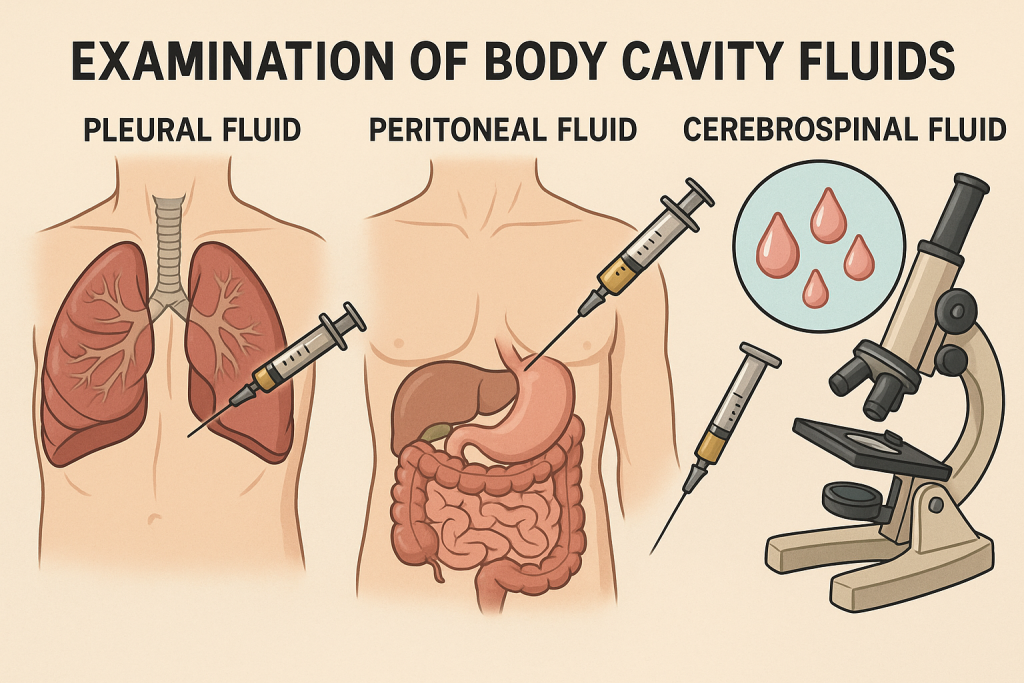
EXAMINATION OF BODY CAVITY FLUIDS
1. Definition:
Body cavity fluids are serous fluids collected from normally closed body cavities. The main types include:
- Pleural fluid – from the pleural (lung) cavity
- Peritoneal fluid (Ascitic fluid) – from the abdominal cavity
- Pericardial fluid – from the sac surrounding the heart
- Synovial fluid – from joint cavities
- Cerebrospinal fluid (CSF) – from the subarachnoid space around the brain and spinal cord (sometimes grouped separately)
2. Indications for Examination:
- Unexplained fever, chest pain, abdominal distention, or joint swelling
- Diagnosis of infections, malignancies, trauma, autoimmune disorders
- Monitoring response to treatment
- Evaluation of transudate vs. exudate
3. Collection of Fluid:
| Fluid Type | Procedure | Site | Needle Insertion |
|---|---|---|---|
| Pleural Fluid | Thoracentesis | Posterior chest wall (between ribs) | Intercostal space |
| Peritoneal Fluid | Paracentesis | Lower abdomen (usually left) | Midline or lateral |
| Pericardial Fluid | Pericardiocentesis | Below xiphoid process or intercostal | Under imaging |
| Synovial Fluid | Arthrocentesis | Affected joint | Directly into joint |
| CSF | Lumbar puncture | Lumbar spine (L3-L4/L4-L5) | Spinal canal |
Note: Aseptic technique and local anesthesia are used; samples are sent in sterile containers to lab.
4. Physical Examination of Fluids:
| Feature | Observations |
|---|---|
| Color | Clear, straw, bloody, cloudy, chylous, greenish |
| Clarity | Transparent, turbid, milky, purulent |
| Volume | Measured in mL |
| Viscosity (for synovial) | String test (normal forms long string) |
| Odor | Foul smell may indicate anaerobic infection |
5. Biochemical Analysis:
| Test | Purpose |
|---|---|
| Protein | Differentiates transudate (<3g/dL) vs. exudate (>3g/dL) |
| Glucose | ↓ in infections (TB, bacterial), RA |
| LDH | ↑ in infections, malignancy |
| pH | ↓ in infection, cancer |
| Amylase | ↑ in pancreatitis or GI perforation (esp. in pleural/peritoneal) |
| Albumin Gradient (SAAG) | >1.1 = portal hypertension; <1.1 = exudate |
6. Microscopic Examination:
| Test | What It Shows |
|---|---|
| Cell count | WBC, RBC – ↑ WBC = infection, malignancy |
| Differential count | Neutrophils (acute), Lymphocytes (TB, cancer) |
| Cytology | Malignant cells in cancers |
| Gram stain | Bacteria presence (helps guide antibiotics) |
| AFB stain | For tuberculosis (Ziehl-Neelsen stain) |
| Culture and Sensitivity | Identifies organisms and antibiotic sensitivity |
| Crystals (Synovial fluid) | Gout (uric acid crystals), Pseudogout (calcium pyrophosphate) |
7. Special Tests:
- ADA (Adenosine Deaminase): High in TB effusion
- PCR/Genetic testing: For TB, viral, or autoimmune markers
- Tumor markers: CA-125 (ovarian), CEA (GI cancers), etc.
- Rheumatoid factor, ANA: In autoimmune effusions
8. Interpretation – Transudate vs. Exudate:
| Feature | Transudate | Exudate |
|---|---|---|
| Protein | <3 g/dL | >3 g/dL |
| LDH | <200 IU/L | >200 IU/L |
| Specific Gravity | <1.015 | >1.015 |
| SAAG | >1.1 g/dL | <1.1 g/dL |
| Causes | CHF, nephrotic syndrome, cirrhosis | TB, cancer, pneumonia, pancreatitis |
9. Nursing Considerations:
- Explain the procedure to the patient
- Ensure consent and prepare sterile equipment
- Position patient appropriately (e.g., sitting for thoracentesis)
- Monitor for complications: hypotension, bleeding, pneumothorax, infection
- Label and send samples to lab promptly
- Provide post-procedure care: rest, monitor vitals, watch for complications
10. Complications of Procedures:
- Hemorrhage
- Infection
- Pneumothorax (in thoracentesis)
- Perforation of organs
- Hypotension
- Pain or discomfort
- Fluid leak (at puncture site)
CEREBROSPINAL FLUID (CSF): METHODS OF COLLECTION & EXAMINATION.
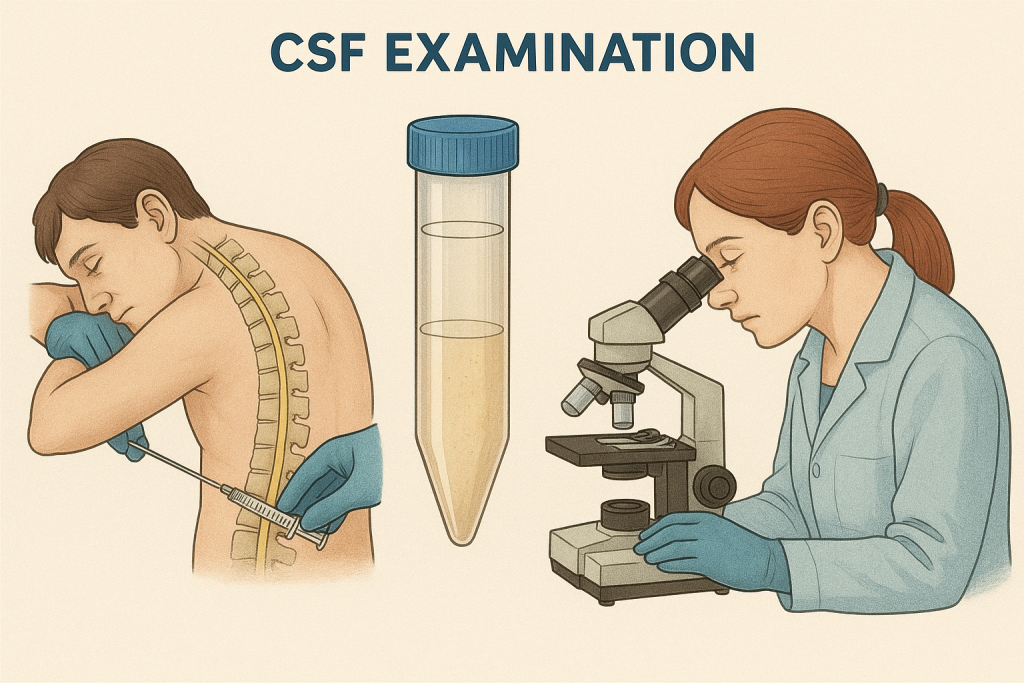
1. What is CSF?
Cerebrospinal Fluid (CSF) is a clear, colorless fluid that surrounds the brain and spinal cord. It cushions the brain, removes waste, and circulates nutrients and hormones.
2. Indications for CSF Examination:
- Suspected meningitis (bacterial, viral, fungal, TB)
- Subarachnoid hemorrhage
- Multiple sclerosis, Guillain-Barré syndrome
- Malignancies (CNS tumors, metastatic spread)
- Neurological symptoms with unknown cause (e.g., seizures, altered mental status)
3. Methods of CSF Collection
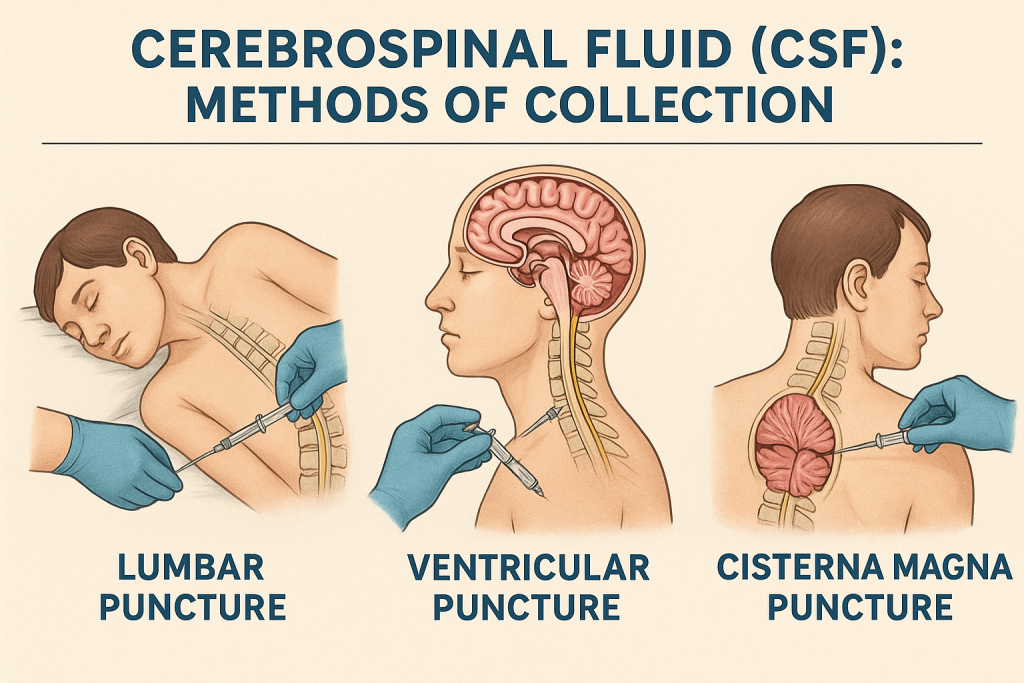
A. Lumbar Puncture (Spinal Tap) – Most Common Method
✅ Procedure Overview:
| Aspect | Details |
|---|---|
| Patient Position | Lateral decubitus (fetal) or sitting position with back curved |
| Site | Lumbar region (usually L3-L4 or L4-L5 interspace) |
| Needle | Lumbar puncture needle with stylet |
| Volume Collected | 3–10 mL (divided into 3–4 sterile tubes) |
| Aseptic Technique | Strictly maintained |
| Contraindications | Increased intracranial pressure (risk of brain herniation), coagulopathy, infection at puncture site |
✅ Steps:
- Explain procedure and obtain informed consent.
- Position the patient correctly.
- Prepare and drape the lumbar area using aseptic technique.
- Insert needle between L3–L4 or L4–L5 vertebrae.
- Withdraw stylet; CSF flows out under normal pressure.
- Collect CSF in 3–4 sterile tubes (1–2 mL each).
- Withdraw needle, apply sterile dressing.
- Instruct patient to lie flat for 1–2 hours.
4. CSF Sample Handling:
| Tube No. | Purpose |
|---|---|
| Tube 1 | Chemistry & glucose |
| Tube 2 | Microbiology (Gram stain, culture) |
| Tube 3 | Cell count & differential |
| Tube 4 | Special tests (PCR, serology, cytology, etc.) |
🚨 Always label tubes properly and send to lab immediately.
5. CSF Examination – Components
A. Physical Examination
| Parameter | Normal | Abnormal Findings |
|---|---|---|
| Color | Clear, colorless | Cloudy (infection), bloody (hemorrhage), xanthochromic (old bleed) |
| Clarity | Transparent | Turbid (infection), oily (contrast), milky (TB/fungal) |
| Opening Pressure | 90–180 mm H₂O (lying down) | ↑ in meningitis, ↓ in shock |
| Volume | Total ~150 mL in body | Normally 3–10 mL withdrawn |
B. Chemical Analysis
| Test | Normal Range | Clinical Significance |
|---|---|---|
| Glucose | 45–80 mg/dL | ↓ in bacterial/TB/fungal meningitis |
| Protein | 15–45 mg/dL | ↑ in infection, MS, tumor, GBS |
| Chloride | 118–132 mEq/L | ↓ in TB meningitis |
| Lactate | <2.1 mmol/L | ↑ in bacterial meningitis |
| Albumin, Globulin | Normal trace | ↑ in multiple sclerosis, infection |
C. Microscopic Examination
| Test | Normal | Abnormal Findings |
|---|---|---|
| WBC count | 0–5 lymphocytes/mm³ | ↑ in infection, inflammation |
| RBC count | 0 | ↑ in hemorrhage or traumatic tap |
| Differential | Lymphocytes | Neutrophils (bacterial), Lymphocytes (viral/TB) |
| Cytology | No abnormal cells | Malignant cells (carcinomatous meningitis) |
D. Microbiological Examination
| Test | Purpose |
|---|---|
| Gram Stain | Rapid detection of bacteria |
| AFB Stain (Ziehl-Neelsen) | Detects Mycobacterium tuberculosis |
| India Ink | Detects Cryptococcus neoformans (fungus) |
| Culture | Gold standard for identifying organisms |
| PCR/NAAT | For TB, herpes virus, CMV, enteroviruses |
E. Other Special Tests:
- Oligoclonal bands: Positive in multiple sclerosis
- VDRL test: Neurosyphilis
- Cryptococcal antigen: Fungal meningitis
- CSF IgG Index: Autoimmune diseases
- Tumor markers: In CNS metastasis
6. Interpretation of CSF Findings (Simplified Table):
| Condition | Pressure | Glucose | Protein | WBC Type |
|---|---|---|---|---|
| Bacterial Meningitis | ↑ | ↓↓ | ↑↑ | Neutrophils ↑ |
| Viral Meningitis | Normal/↑ | Normal | Mild ↑ | Lymphocytes ↑ |
| Tubercular Meningitis | ↑ | ↓ | ↑ | Lymphocytes ↑ |
| Fungal Meningitis | ↑ | ↓ | ↑ | Lymphocytes ↑ |
| Subarachnoid Hemorrhage | ↑ | Normal | ↑ | RBC ↑, Xanthochromia |
7. Nursing Responsibilities:
- Pre-procedure:
- Explain procedure and reduce anxiety.
- Obtain consent.
- Ensure patient has voided before procedure.
- During:
- Assist with patient positioning.
- Maintain strict asepsis.
- Monitor for pain, dizziness, or complications.
- Post-procedure:
- Keep patient flat (supine) for at least 1–2 hours.
- Monitor vitals and watch for headache or CSF leak.
- Encourage fluid intake.
- Send samples promptly with correct labeling.
8. Complications of Lumbar Puncture:
- Post-lumbar puncture headache (common)
- Bleeding or hematoma
- Infection
- Brain herniation (if ICP elevated)
- Nerve damage (rare)
- CSF leak
SPUTUM COLLECTION AND EXAMINATION
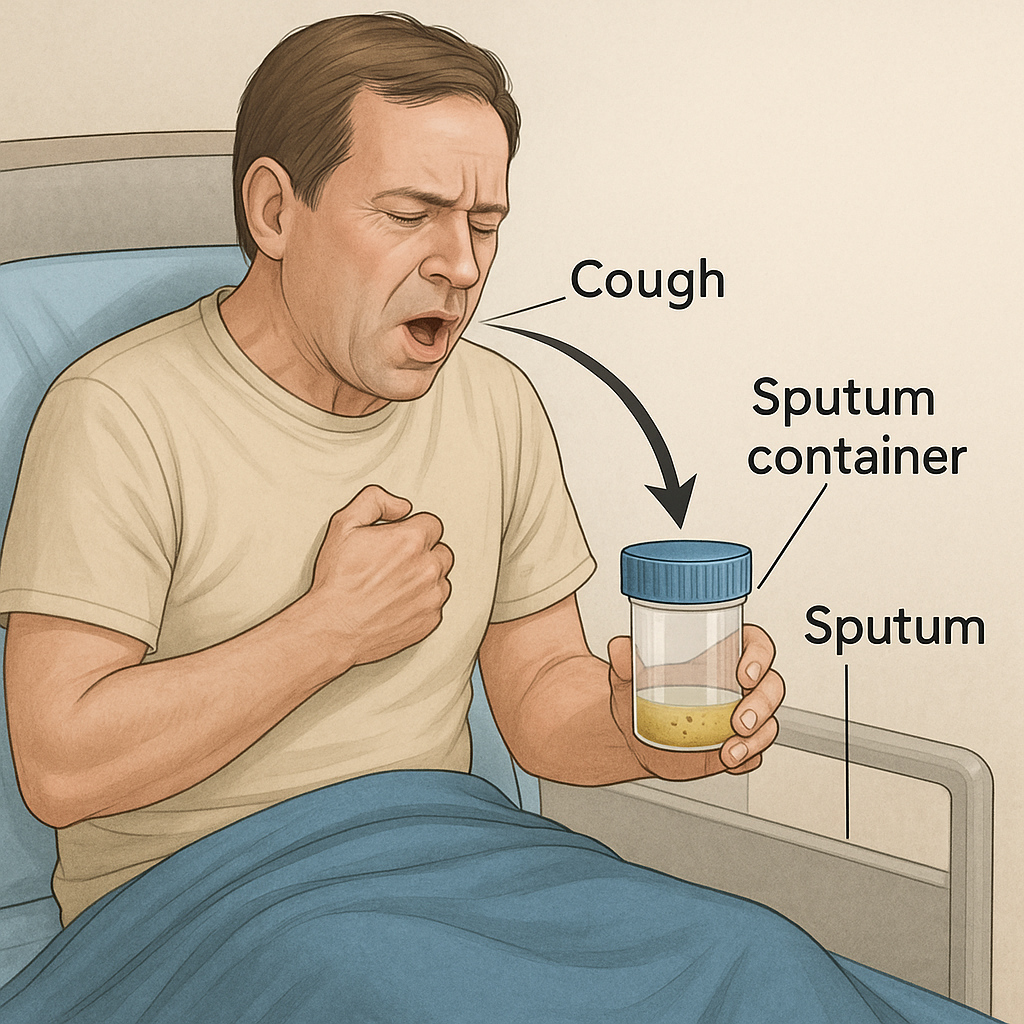
1. What is Sputum?
Sputum is the thick mucus or phlegm that is coughed up from the lower respiratory tract (lungs and bronchi), not to be confused with saliva. It is analyzed to diagnose respiratory diseases such as tuberculosis, pneumonia, asthma, bronchitis, lung cancer, and others.
2. Purposes of Sputum Examination:
- Diagnose infections (e.g., TB, pneumonia, bronchitis, fungal infection)
- Detect malignant cells (lung cancer)
- Identify allergic conditions (eosinophilia in asthma)
- Monitor treatment response
- Investigate hemoptysis (coughing blood)
3. Methods of Sputum Collection
There are three main methods based on patient condition and sputum availability:
A. Spontaneous (Self-expectorated) Sputum Collection
Most common method
Instructions to patient:
- Best done early morning before eating or drinking.
- Rinse mouth with water (not antiseptic) to remove food particles and oral bacteria.
- Take deep breaths and cough forcefully from deep in the chest.
- Collect 5–10 mL sputum in a sterile, wide-mouthed, screw-capped container.
- Ensure it is sputum (thick, mucoid) and not saliva (thin, watery).
B. Induced Sputum Collection
For patients unable to cough up sputum spontaneously
Procedure:
- Patient inhales hypertonic saline (3–5%) via nebulizer for 10–20 minutes.
- This loosens secretions and induces coughing.
- Sputum is then collected in sterile container.
C. Suctioned (Invasive) Sputum Collection
Used for critically ill, unconscious, or ventilated patients.
Methods include:
- Oropharyngeal or nasopharyngeal suction
- Endotracheal or tracheostomy suctioning
Using a sterile suction catheter, connected to vacuum apparatus.
4. Precautions During Collection:
- Use sterile containers.
- Ensure adequate labeling (patient name, date, time).
- Avoid contamination with saliva.
- Wear gloves, mask, and gown (especially for TB cases).
- Collect in a well-ventilated or isolation room if infection is suspected.
5. Physical Examination of Sputum:
| Characteristic | Findings | Interpretation |
|---|---|---|
| Amount | Small, moderate, large | Large = infection or pulmonary edema |
| Color | Clear, white, yellow, green, rusty, pink | Green/yellow = infection, Rusty = pneumonia, Pink frothy = pulmonary edema |
| Consistency | Mucoid, purulent, frothy, bloody | Purulent = infection, Bloody = TB or cancer |
| Odor | Foul-smelling | Anaerobic infections |
| Presence of blood | Hemoptysis | TB, cancer, trauma |
6. Laboratory Examination of Sputum:
A. Microscopic Examination
1. Gram Stain:
- Identifies bacteria (gram-positive/negative)
- Guides initial antibiotic therapy
2. Acid-Fast Bacilli (AFB) Stain – Ziehl-Neelsen:
- For Tuberculosis diagnosis
- 3 early morning samples on consecutive days are preferred
3. India Ink Preparation:
- Detects Cryptococcus neoformans (fungal infection)
4. Wet Mount or KOH Test:
- For fungal elements
5. Cytology:
- Identifies malignant cells in lung cancer or metastasis
6. Eosinophil Count:
- ↑ in asthma, parasitic infections, allergic bronchitis
B. Culture and Sensitivity (C&S):
- Sample is cultured on specific media (Blood agar, MacConkey, Lowenstein-Jensen for TB, Sabouraud agar for fungi).
- Identifies bacteria/fungi and tests for antibiotic sensitivity.
- Takes 2–3 days for bacteria, up to 6–8 weeks for TB culture.
C. Molecular Tests:
1. CBNAAT / GeneXpert:
- Rapid test for Mycobacterium tuberculosis and Rifampicin resistance.
- Results within 2 hours.
2. PCR-based Tests:
- Highly sensitive for viruses, TB, and certain fungi.
7. Interpretation of Results:
| Finding | Suggestive Diagnosis |
|---|---|
| Yellow/green, purulent with neutrophils and gram-positive cocci | Bacterial pneumonia |
| Blood-streaked sputum with AFB positive | Tuberculosis |
| Rusty sputum | Pneumococcal pneumonia |
| Foul-smelling, thick sputum with mixed flora | Anaerobic lung abscess |
| Pink frothy sputum | Pulmonary edema |
| Malignant cells on cytology | Lung cancer |
8. Nursing Responsibilities:
- Explain procedure and importance of proper collection
- Ensure privacy and comfort
- Use PPE (especially for TB patients)
- Encourage deep breathing before coughing
- Label and send samples to lab immediately
- Document the color, consistency, and amount of sputum
- Monitor for signs of respiratory distress during collection
9. Common Errors to Avoid:
- Collecting saliva instead of sputum
- Delayed transport to lab (may kill pathogens)
- Using non-sterile containers
- Not instructing patient properly
WOUND DISCHARGE (EXUDATE): COLLECTION AND EXAMINATION
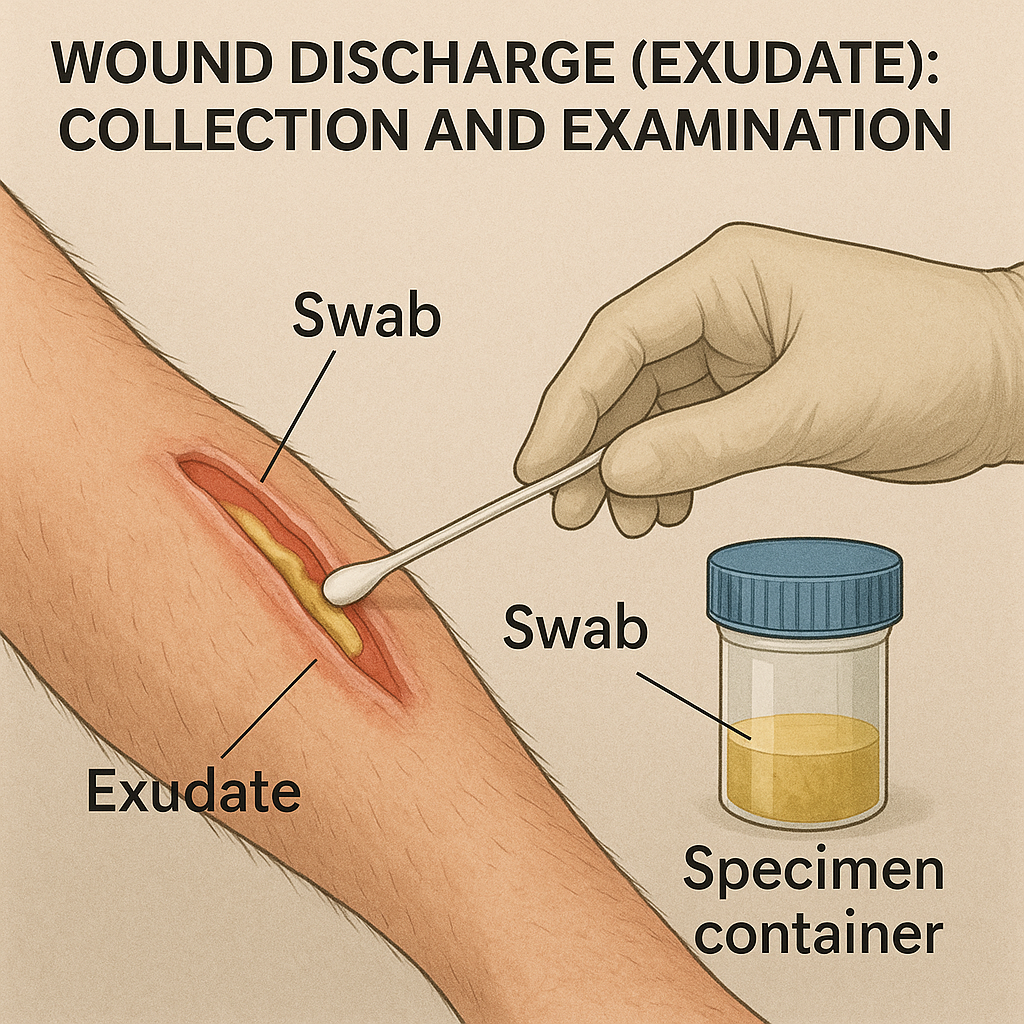
1. What is Wound Discharge?
Wound discharge, or wound exudate, is the fluid that leaks from wounds. It contains plasma, white blood cells, dead cells, bacteria, and proteins, and its nature gives vital clues about the type of infection, healing status, or complications.
2. Purposes of Wound Discharge Examination:
- Identify type of infection (bacterial, fungal, MRSA, etc.)
- Determine healing stage of the wound
- Diagnose resistant organisms
- Guide antibiotic therapy
- Assess for malignancy in chronic wounds
3. Types of Wound Discharge (Clinically Observed):
| Type | Description | Indication |
|---|---|---|
| Serous | Clear, watery | Normal healing |
| Sanguineous | Bright red, bloody | Trauma, bleeding |
| Serosanguineous | Pale red, watery | Normal early healing |
| Purulent | Thick, yellow/green | Infection |
| Foul-smelling | May be green/brown | Anaerobic infection |
| Seropurulent | Watery + pus | Early/mild infection |
4. Methods of Collection of Wound Discharge
A. Swab Technique (Most Common)
Used for superficial wounds or small ulcers.
Steps:
- Clean wound with sterile saline (to remove surface contaminants).
- Use a sterile cotton swab.
- Rotate the swab gently over the wound base, targeting areas with visible pus or tissue slough (not just surface).
- Place the swab into a sterile transport tube with medium (like Amies medium).
- Label and send immediately to lab.
Note: Avoid swabbing dry or necrotic tissue.
B. Aspirate Collection (Wound Aspiration)
Used for deep or closed wounds, abscesses, or pockets.
Steps:
- Clean skin with antiseptic.
- Use a sterile syringe and needle.
- Insert into the wound pocket and aspirate fluid.
- Transfer into a sterile container or culture bottle.
C. Tissue Biopsy (In Special Cases)
Used when infection is deep, chronic, or unresponsive to treatment.
Steps:
- Done by a physician or trained personnel.
- A small piece of wound tissue is surgically removed.
- Sent for histopathology, culture, and cytology.
5. Transport & Lab Handling:
- Transport sample within 1–2 hours of collection.
- Avoid refrigeration unless instructed by the lab.
- Ensure correct labeling: patient name, date, wound site, clinical suspicion.
6. Laboratory Examination of Wound Discharge
A. Macroscopic (Physical) Examination:
| Feature | Observation | Significance |
|---|---|---|
| Color | Yellow, green, red, brown | Green = Pseudomonas, Yellow = Staph, Brown = anaerobes |
| Consistency | Thin, thick, foul-smelling | Thick + foul = bacterial infection |
| Odor | Sweet, putrid | Sweet = Pseudomonas, Putrid = anaerobes |
B. Microscopic Examination:
| Test | Purpose |
|---|---|
| Gram Stain | Detects gram-positive or gram-negative bacteria, pus cells |
| KOH Mount | For fungal infections (Candida, Aspergillus) |
| AFB Stain (Ziehl-Neelsen) | Detects Mycobacterium tuberculosis in chronic wounds |
| Wet Mount | Parasitic or fungal elements if suspected |
C. Culture and Sensitivity (C&S):
Purpose: To grow and identify the pathogen and determine its antibiotic sensitivity.
- Media used: Blood agar, MacConkey agar, Chocolate agar, Sabouraud’s (for fungi).
- Culture incubated 24–72 hours.
- Report shows:
- Organism identified
- Sensitive/Resistant antibiotics
D. Special Investigations:
| Test | Use |
|---|---|
| MRSA Screening | Detect Methicillin-resistant Staphylococcus aureus |
| PCR Testing | For resistant genes, TB |
| Cytology | If malignancy suspected in chronic ulcers |
| Biopsy and Histopathology | For cancer, chronic granulomatous infection |
7. Interpretation of Results:
| Finding | Suggestive Diagnosis |
|---|---|
| Pus cells + gram-positive cocci | Staph aureus infection |
| Pus cells + gram-negative rods | Pseudomonas, E. coli |
| Green discharge + sweet odor | Pseudomonas infection |
| Foul-smelling discharge + gas bubbles | Anaerobic infection |
| Chronic non-healing ulcer + abnormal cells | Malignant transformation (Marjolin’s ulcer) |
8. Nursing Responsibilities:
- Educate the patient about wound hygiene
- Use sterile technique during collection
- Ensure correct labeling and documentation
- Wear PPE during procedure
- Monitor for local signs of infection (redness, warmth, pain, swelling)
- Assist in wound cleaning and dressing post-collection
- Report lab results to medical team promptly
9. Common Errors to Avoid:
- Swabbing before cleaning wound
- Collecting from slough or necrotic tissue
- Delayed transport to lab
- Using dry swab (use pre-moistened if needed)
- Poor labeling
EXAMINATION OF BODY CAVITY FLUIDS
👉 Including: CSF, Sputum, Wound Discharge
I. CEREBROSPINAL FLUID (CSF)
A. Collection Method – Lumbar Puncture
| Feature | Details |
|---|---|
| Site | L3–L4 or L4–L5 interspace |
| Position | Lateral recumbent or sitting |
| Volume | 3–10 mL (divided into 3–4 tubes) |
| Sterility | Strict aseptic technique |
| Contraindications | Increased intracranial pressure, bleeding disorders |
B. Examination of CSF
| Test Type | Tests & Purpose |
|---|---|
| Physical | Color, clarity, pressure, volume |
| Biochemistry | Glucose, protein, chloride, lactate |
| Clinical Pathology | Cell count, differential count |
| Microbiology | Gram stain, AFB stain, India ink, culture, PCR (GeneXpert for TB, viruses), fungal studies |
| Special Tests | Cytology (for malignancy), oligoclonal bands (MS), VDRL (syphilis) |
II. SPUTUM (Respiratory Tract Secretion)
A. Collection Methods
| Method | Description |
|---|---|
| Spontaneous expectoration | Early morning, after rinsing mouth |
| Induced sputum | Nebulized hypertonic saline inhalation |
| Suction method | In ventilated or unconscious patients (tracheal aspiration or BAL) |
B. Examination of Sputum
| Test Type | Tests & Purpose |
|---|---|
| Physical | Color, odor, volume, consistency |
| Biochemistry | Not commonly done unless for eosinophils in asthma |
| Clinical Pathology | Cytology (malignant cells), eosinophil count |
| Microbiology | Gram stain, AFB stain (for TB), KOH mount (fungi), Culture & Sensitivity, CBNAAT/GeneXpert, PCR (for viruses, TB) |
III. WOUND DISCHARGE (Exudate or Pus)
A. Collection Methods
| Method | Procedure |
|---|---|
| Swab technique | After cleaning wound, rotate sterile swab over base of wound |
| Aspirate | Using sterile syringe to aspirate pus from deeper tissue |
| Tissue biopsy | In chronic wounds or for malignancy detection |
B. Examination of Wound Discharge
| Test Type | Tests & Purpose |
|---|---|
| Physical | Color, consistency, odor, amount |
| Biochemistry | Rarely performed unless for special wound healing studies |
| Clinical Pathology | Cytology (to detect cancer cells in chronic wounds) |
| Microbiology | Gram stain, AFB stain, KOH mount, Culture & Sensitivity, MRSA screening, PCR for resistant genes |
IV. EXAMINATION PARAMETERS AT A GLANCE
| Specimen | Physical | Clinical Pathology | Biochemistry | Microbiology |
|---|---|---|---|---|
| CSF | Color, clarity, pressure | Cell count, cytology | Glucose, protein, chloride, lactate | Gram stain, AFB, culture, PCR |
| Sputum | Color, odor, consistency | Eosinophils, cytology | Rarely done | Gram stain, AFB, fungal stains, culture |
| Wound Discharge | Color, odor, type | Cytology | Rarely done | Gram stain, culture, AFB, fungal tests |
V. General Guidelines for Sample Collection
- Always use sterile containers and aseptic technique
- Collect adequate volume
- Label all specimens with patient name, date, time, and source
- Transport to lab immediately to avoid contamination or degradation
- Wear PPE when handling infectious samples
- Inform lab of any clinical suspicion (e.g., TB, fungal, malignancy)
VI. Common Pathogens Detected in Each Fluid
| Fluid | Common Pathogens |
|---|---|
| CSF | Streptococcus pneumoniae, Neisseria meningitidis, Mycobacterium tuberculosis, HSV, Cryptococcus |
| Sputum | Mycobacterium tuberculosis, Klebsiella, Streptococcus pneumoniae, Pseudomonas, Candida |
| Wound Discharge | Staphylococcus aureus (including MRSA), Streptococcus, Pseudomonas, anaerobes, TB, fungi |
VII. Nursing Responsibilities
- Explain the procedure and prepare the patient
- Maintain strict aseptic technique during collection
- Properly label and transport samples
- Document relevant clinical details
- Observe patient for complications (e.g., headache after LP, respiratory distress during sputum collection)
SEMEN ANALYSIS
1. Definition:
Semen analysis is a laboratory test to assess the quantity and quality of semen and sperm to determine male fertility potential or diagnose conditions affecting sperm production.
2. Purposes / Indications:
- Evaluation of male infertility
- After vasectomy (to confirm absence of sperm)
- Investigation of urological or endocrine disorders
- Assessment of sperm count, motility, and morphology
- Donor sperm screening
3. Preparation for the Test:
| Instruction | Duration |
|---|---|
| Abstinence | 2–7 days before the test (avoid ejaculation) |
| No alcohol/smoking | 48–72 hours prior |
| Avoid hot baths/saunas | 2–5 days prior |
| Avoid medications | Antibiotics, steroids, hormones (if advised by doctor) |
4. Method of Collection:
| Step | Details |
|---|---|
| Sample Collection | By masturbation directly into a sterile, wide-mouthed container |
| Environment | Private room (clinic/lab) or at home (delivered within 30–60 mins) |
| Container | Sterile, non-toxic plastic container (no condoms unless approved for sperm collection) |
| Temperature | Kept at body temperature (~37°C) during transport |
🚨 Avoid contamination with lubricants, saliva, water, or toilet paper.
5. Time for Testing:
- The sample should be tested within 1 hour of collection for accurate motility and morphology evaluation.
6. Semen Analysis Parameters:
Based on WHO (2021) Reference Values
A. Physical Characteristics
| Parameter | Normal Value |
|---|---|
| Volume | 1.5 mL or more |
| Color | Whitish-gray |
| Viscosity | Liquefied within 30 mins |
| pH | 7.2–8.0 |
| Odor | Distinctive chlorine-like |
| Liquefaction time | Within 15–30 minutes |
B. Microscopic Analysis
| Parameter | Normal Range | Significance |
|---|---|---|
| Sperm Count (Concentration) | ≥15 million/mL | ↓ = oligospermia; 0 = azoospermia |
| Total Sperm Count | ≥39 million/ejaculate | Indicates fertility potential |
| Motility (progressive) | ≥32% progressively motile | ↓ = asthenozoospermia |
| Total Motility (all moving) | ≥40% (progressive + non-progressive) | |
| Vitality (Live sperm) | ≥58% live | Dead sperm = necrozoospermia |
| Morphology (Normal forms) | ≥4% (strict criteria) | Abnormal = teratozoospermia |
| WBCs | <1 million/mL | ↑ = infection or inflammation |
C. Additional Tests (If Indicated)
| Test | Purpose |
|---|---|
| Fructose Test | Confirms seminal vesicle function (low = ejaculatory duct obstruction) |
| MAR or IgG Antisperm Antibody Test | Detects antisperm antibodies |
| Sperm DNA Fragmentation Test | Measures sperm genetic integrity |
| Culture and Sensitivity | Identifies infections |
| Hormonal Assays | FSH, LH, testosterone in abnormal cases |
| Ultrasound | For varicocele, obstruction, tumors |
7. Interpretation of Common Findings
| Result | Interpretation |
|---|---|
| Low volume + acidic pH | Ejaculatory duct obstruction |
| No sperm + normal volume | Azoospermia (obstructive or non-obstructive) |
| Low motility + abnormal shape | Likely infertility |
| High WBC count | Seminal infection |
| Low fructose | Ejaculatory dysfunction or congenital absence of vas deferens |
8. Nursing/Collection Responsibilities:
- Explain purpose and procedure clearly
- Ensure patient privacy and comfort
- Provide labeled sterile container
- Educate on abstinence and sample transport
- Coordinate prompt delivery to lab
- Document relevant medical or medication history
9. Repeat Testing:
- At least 2–3 samples, spaced 2–3 weeks apart, are recommended due to natural sperm fluctuations.
10. Clinical Conditions Diagnosed via Semen Analysis:
- Infertility (primary or secondary)
- Azoospermia (no sperm)
- Oligospermia (low count)
- Asthenozoospermia (poor motility)
- Teratozoospermia (abnormal shape)
- Varicocele
- Obstructive pathologies
- Hormonal disorders
SEMEN ANALYSIS
1. Introduction
Semen analysis is the cornerstone of evaluating male fertility. The quality of sperm is assessed by analyzing:
- Sperm Count – the number of sperm
- Sperm Motility – the ability to move
- Sperm Morphology – the shape and structure
All three parameters must be evaluated together to assess a man’s fertility potential.
2. SPERM COUNT (Sperm Concentration)
Definition:
The number of spermatozoa present in 1 milliliter (mL) of semen.
Normal Range (WHO 2021):
- ≥15 million sperm/mL
- ≥39 million per ejaculate (total sperm count)
Clinical Interpretation:
| Count | Term | Significance |
|---|---|---|
| ≥15 million/mL | Normal | Good fertility potential |
| <15 million/mL | Oligospermia | Reduced fertility |
| 0 sperm | Azoospermia | Complete absence of sperm; may indicate blockage or failure of sperm production |
Importance in Infertility:
- Low sperm count reduces the chance of sperm reaching the egg.
- A very low or zero count often necessitates further testing (e.g., hormonal profile, testicular biopsy, imaging).
3. SPERM MOTILITY
Definition:
The ability of sperm to move forward (progressive movement) through the female reproductive tract to reach the egg.
Types of Motility:
| Type | Description |
|---|---|
| Progressive | Moves actively in a straight line or large circles |
| Non-progressive | Moves but without forward progression |
| Immotile | No movement |
Normal Values (WHO 2021):
- Progressive motility: ≥32%
- Total motility (progressive + non-progressive): ≥40%
Clinical Interpretation:
| Condition | Description |
|---|---|
| Asthenozoospermia | Reduced sperm motility |
| Total immotility | Associated with infertility; may indicate genetic or structural defects |
Importance in Infertility:
- Without adequate motility, sperm cannot reach or penetrate the egg.
- Poor motility significantly decreases the chances of natural conception.
4. SPERM MORPHOLOGY
Definition:
The study of sperm shape and structure, including:
- Head (contains DNA)
- Midpiece (energy production)
- Tail (movement)
Normal Value (WHO 2021 – Strict Kruger Criteria):
- ≥4% normal forms
(Only 4% or more of sperm need to have a normal shape for the sample to be considered normal)
Abnormalities Include:
| Area | Abnormalities |
|---|---|
| Head | Large, small, double heads, amorphous |
| Midpiece | Thick, irregular |
| Tail | Coiled, multiple tails, short tail |
Clinical Interpretation:
| Term | Description |
|---|---|
| Teratozoospermia | High % of abnormally shaped sperm |
| Normal morphology with low count/motility | Still may result in infertility |
Importance in Infertility:
- Abnormal sperm may not penetrate the egg.
- Poor morphology is linked to failed fertilization in natural and assisted reproduction.
5. Summary Table – Normal Values & Significance
| Parameter | WHO Normal Value | Importance |
|---|---|---|
| Sperm Count | ≥15 million/mL | Fertility potential; low count = reduced chance |
| Motility | ≥40% total; ≥32% progressive | Required for sperm to reach egg |
| Morphology | ≥4% normal forms | Required for fertilization |
6. Role in Infertility Diagnosis
- A man with abnormal values in one or more parameters may have subfertility or infertility.
- Causes of abnormalities may include:
- Varicocele
- Hormonal imbalances
- Infections
- Genetic conditions
- Environmental/lifestyle factors (smoking, alcohol, heat exposure)
- Repeat testing and further investigations like hormonal profile, scrotal ultrasound, genetic testing, or testicular biopsy may be needed.
URINE EXAMINATION

I. PHYSICAL CHARACTERISTICS OF URINE
These are macroscopic observations made immediately after collection:
| Characteristic | Normal Findings | Clinical Significance of Abnormalities |
|---|---|---|
| Color | Pale yellow to amber | Red (hematuria), brown (bilirubin), cloudy (infection), dark yellow (dehydration) |
| Clarity | Clear | Turbid/cloudy in infection, crystals, mucus |
| Odor | Mild, aromatic | Foul (infection), fruity (ketones/diabetes), ammonia (old sample) |
| Volume | 1–2 liters/day (normal adult) | Oliguria (<400 mL/day), Polyuria (>2.5 L/day), Anuria (<100 mL/day) |
| Specific Gravity | 1.005–1.030 | Low = dilute (diabetes insipidus); High = dehydration, proteinuria |
| pH | 4.5–8.0 | Acidic in diabetes, starvation; Alkaline in UTI, vegetarian diet |
II. URINE ANALYSIS (Routine/Microscopic/Biochemical)
A. Chemical (Dipstick) Analysis:
| Parameter | Normal | Abnormal Findings |
|---|---|---|
| Protein | Negative | Proteinuria – kidney disease, preeclampsia |
| Glucose | Negative | Glycosuria – diabetes mellitus |
| Ketones | Negative | Ketosis – diabetes, starvation |
| Blood | Negative | Hematuria – UTI, stones, trauma |
| Leukocyte esterase | Negative | Indicates WBCs – infection |
| Nitrites | Negative | Positive = gram-negative bacterial UTI |
| Bilirubin | Negative | Hepatic disease |
| Urobilinogen | Normal trace | Increased in hemolysis, liver dysfunction |
| pH & SG | As above | Evaluates acid-base and concentrating ability |
B. Microscopic Examination (Centrifuged Sediment):
| Element | Normal | Clinical Significance |
|---|---|---|
| RBCs | 0–2/HPF | Hematuria – trauma, stones, glomerulonephritis |
| WBCs | 0–5/HPF | Pyuria – UTI |
| Epithelial Cells | Few | Many = contamination or kidney disease |
| Casts | Occasional hyaline | RBC casts – glomerulonephritis, WBC casts – pyelonephritis |
| Crystals | Occasional | Excessive – kidney stones, metabolic disorder |
| Bacteria/Yeast | None | Infection, contamination |
III. URINE CULTURE AND SENSITIVITY (C&S)
Used to identify bacterial or fungal infections in the urinary tract and test which antibiotics are effective.
A. Specimen Collection:
| Type | Method |
|---|---|
| Midstream Clean-Catch | Most common; for adults |
| Catheterized sample | In catheterized patients |
| Suprapubic aspiration | In infants or sterile collection needs |
| Pediatric urine bag | For infants (non-invasive) |
Container: Sterile, leak-proof container
Timing: Transport to lab within 1 hour or refrigerate up to 24 hours
B. Procedure in Lab:
- A small amount of urine is plated on culture media (blood agar, MacConkey agar).
- Plates are incubated at 37°C for 18–24 hours.
- Colony count is done to determine significance:
- >10⁵ CFU/mL → Significant bacteriuria (infection)
- 10³–10⁴ CFU/mL → Possible infection (repeat if asymptomatic)
- <10³ CFU/mL → Contamination likely
- Identified organisms are tested for antibiotic sensitivity using:
- Kirby-Bauer Disc Diffusion
- MIC testing (Minimum Inhibitory Concentration)
C. Common Uropathogens Identified:
| Organism | Common Infections |
|---|---|
| Escherichia coli | Most common UTI |
| Klebsiella pneumoniae | Nosocomial UTI |
| Proteus mirabilis | Alkaline urine, stones |
| Enterococcus faecalis | Complicated UTI |
| Staphylococcus saprophyticus | Young women, honeymoon cystitis |
| Candida albicans | Diabetics, catheterized, immunocompromised |
IV. Clinical Relevance in Diagnosing Conditions:
| Finding | Associated Condition |
|---|---|
| Proteinuria + RBC casts | Glomerulonephritis |
| Pyuria + WBC casts | Pyelonephritis |
| Nitrites + leukocyte esterase | Bacterial UTI |
| Glycosuria + ketonuria | Uncontrolled diabetes mellitus |
| Hematuria | Stones, tumors, trauma |
| Acidic urine | Diabetic ketoacidosis, dehydration |
| Alkaline urine | UTI with Proteus or Klebsiella |
V. Nursing/Collection Guidelines:
- Explain and assist with clean-catch technique
- Use sterile container
- Label with patient name, time, date
- Send sample to lab promptly
- Document medications (e.g., antibiotics), recent symptoms, and fluid intake
- Encourage patient to drink water unless restricted
FAECES (STOOL): CHARACTERISTICS
1. Normal Characteristics of Faeces
| Parameter | Normal Value / Appearance |
|---|---|
| Color | Light to dark brown |
| Consistency | Soft, formed |
| Shape | Cylindrical, sausage-like |
| Amount | ~100–200 grams/day (varies with diet) |
| Frequency | 1–2 times/day to once in 2 days |
| Odor | Slightly offensive, characteristic fecal smell |
| pH | Slightly acidic to neutral (pH 6–7.5) |
| Mucus | Absent or minimal |
| Undigested food | Minimal if digestion is normal |
| Blood | Absent |
| Parasites | None visible or microscopically present |
2. Abnormal Faecal Characteristics & Clinical Significance
| Characteristic | Abnormal Findings | Clinical Indication |
|---|---|---|
| Color | Black, tarry (melena) | Upper GI bleeding (e.g., ulcer, varices) |
| Bright red | Lower GI bleeding (e.g., hemorrhoids, cancer) | |
| Clay/white | Bile duct obstruction (liver/gallbladder disease) | |
| Green | Rapid transit, spinach, antibiotics | |
| Yellow, greasy | Steatorrhea (fatty stool – malabsorption, pancreatitis) | |
| Consistency | Watery | Diarrhea – infection, IBS |
| Hard, dry | Constipation, dehydration | |
| Pasty, foul, bulky | Fat malabsorption, celiac disease | |
| Odor | Foul-smelling | Putrefaction, infection, GI bleeding |
| Acidic odor | Carbohydrate malabsorption (e.g., lactose intolerance) | |
| Mucus | Excess mucus | Inflammatory bowel disease (IBD), infection |
| Blood | Visible red blood | Hemorrhoids, fissures, cancer |
| Occult (hidden) | Detected in fecal occult blood test (FOBT) – GI bleeding | |
| Parasites | Visible worms or eggs | Intestinal parasitic infestation (e.g., roundworms, hookworms) |
| Pus | Present | Dysentery, IBD, abscess |
3. Bristol Stool Chart (Clinical Reference Tool)
Used to assess stool form as an indicator of intestinal transit time:
| Type | Description | Interpretation |
|---|---|---|
| Type 1 | Hard lumps, separate | Severe constipation |
| Type 2 | Lumpy and sausage-like | Mild constipation |
| Type 3 | Sausage-shaped with cracks | Normal |
| Type 4 | Smooth, soft sausage/snake | Ideal, healthy stool |
| Type 5 | Soft blobs with clear edges | Lacking fiber |
| Type 6 | Mushy, fluffy with ragged edges | Mild diarrhea |
| Type 7 | Watery, no solid pieces | Severe diarrhea |
4. Nursing Responsibilities (Stool Observation)
- Observe and document color, consistency, odor, frequency
- Note presence of blood, mucus, pus, undigested food, or worms
- Use standard precautions when handling samples
- Collect sample in clean, dry, labeled container
- Send for lab examination promptly if ordered
- Educate patient on normal vs. abnormal stool
STOOL EXAMINATION

1. PURPOSE OF STOOL EXAMINATION
- Detect digestive system disorders
- Diagnose infections (bacterial, parasitic, fungal)
- Detect GI bleeding, malabsorption, or inflammatory conditions
- Identify parasites and their life stages
- Evaluate enzymatic or carbohydrate digestion problems
2. TYPES OF STOOL EXAMINATION
| Type | Involves |
|---|---|
| Physical Examination | Color, consistency, odor, blood, mucus, parasites |
| Microscopic Examination | Cells, ova, cysts, parasites, crystals |
| Chemical Tests | Occult blood, reducing substances, pH, fats |
| Microbiological Tests | Culture & Sensitivity (bacteria/fungi), PCR |
| Special Tests | Enzyme levels, fat globules, antigen detection |
3. TESTS IN DETAIL
A. OCCULT BLOOD TEST (FOBT – Fecal Occult Blood Test)
Purpose:
Detects hidden (microscopic) blood in stool, used to screen for colon cancer, polyps, and GI bleeding.
Method:
- Guaiac-based test (gFOBT)
- Immunochemical test (iFOBT or FIT)
Instructions:
- Avoid red meat, iron, NSAIDs, vitamin C, aspirin for 2–3 days before the test (for gFOBT)
- Collect a small sample from multiple spots
- Repeat on 2–3 separate days if screening
Interpretation:
| Result | Suggestion |
|---|---|
| Positive | Bleeding from polyps, ulcers, cancer, hemorrhoids, IBD |
| Negative | No detectable blood (may still require further tests) |
B. OVA, PARASITES & CYSTS (O&P Test)
Purpose:
To detect intestinal parasites (protozoa, helminths) and their life stages.
Collection:
- Collect fresh stool sample in clean, dry container.
- For parasites, 3 samples on different days may be needed.
- Avoid contamination with urine or water.
Microscopic Examination:
| Component | Seen in |
|---|---|
| Ova (eggs) | Ascaris, Hookworm, Trichuris, etc. |
| Cysts | Giardia lamblia, Entamoeba histolytica |
| Trophozoites | Active forms of protozoa – must be examined fresh |
| Larvae | Strongyloides, hookworm |
| Worms (whole) | Roundworms, pinworms may be visible |
Saline and iodine wet mounts are used to visualize under microscope.
C. REDUCING SUBSTANCE TEST (Benedict’s Test)
Purpose:
Detects unabsorbed sugars in stool, used in infants to diagnose carbohydrate malabsorption (e.g., lactose intolerance).
Principle:
Reducing sugars (like glucose, galactose) reduce Benedict’s reagent, causing a color change.
Interpretation:
| Result | Meaning |
|---|---|
| Positive | Unabsorbed carbohydrates (lactose intolerance, enzyme deficiency) |
| Negative | Normal sugar absorption |
D. STOOL pH TEST
| pH | Indication |
|---|---|
| <5.5 (acidic) | Carbohydrate malabsorption, bacterial fermentation |
| >7.5 (alkaline) | Bacterial overgrowth, high protein diet |
E. FAT TEST (Sudan III Stain)
Purpose:
Detects undigested fat globules in stool, seen in malabsorption syndromes like:
- Celiac disease
- Pancreatic insufficiency
- Cystic fibrosis
Positive test shows orange-red fat globules under microscope.
4. SUPPORTING MICROBIOLOGICAL EXAMINATIONS
| Test | Purpose |
|---|---|
| Stool Culture | Identifies bacteria (Salmonella, Shigella, E. coli) |
| C. difficile toxin assay | Detects Clostridium difficile infection |
| PCR / ELISA | Detects viral/parasitic DNA or antigens |
| Fungal culture | For immunocompromised patients |
5. INTERPRETATION AT A GLANCE
| Test | Positive Result Indicates |
|---|---|
| Occult Blood | GI bleeding, cancer, ulcer, IBD |
| Ova/Parasites | Intestinal parasitic infection |
| Reducing Substance | Lactose intolerance, enzyme deficiency |
| Fat Globules | Steatorrhea, malabsorption |
| WBCs in stool | Inflammation, infection (dysentery, IBD) |
| Mucus | IBD, infection, IBS |
6. Nursing Responsibilities for Stool Collection:
- Educate patient on collection method (especially for O&P test: no urine/water contamination)
- Use sterile, leak-proof containers
- Label sample with name, date, time
- Transport to lab within 30–60 minutes (especially for parasite or trophozoite identification)
- Wear gloves and follow infection control practices
- Document findings and any patient symptoms (diarrhea, pain, fever)
METHODS AND COLLECTION OF URINE & FAECES FOR VARIOUS TESTS
I. URINE COLLECTION METHODS FOR VARIOUS TESTS
A. Routine Urine Analysis
Purpose:
Detect infections, kidney disorders, glucose, protein, ketones, and other abnormalities.
Method:
- Type: Random or early morning midstream clean-catch sample
- Container: Sterile, wide-mouthed, dry plastic container
- Instructions:
- Wash genital area before collection
- Discard the first stream of urine, collect midstream sample
- Avoid contamination
- Volume Needed: 10–30 mL
B. Urine Culture & Sensitivity (C&S)
Purpose:
Detect and identify organisms (usually bacteria) causing urinary tract infection.
Method:
- Type: Midstream Clean-Catch (MSCC)
- Sterile container with tightly sealed lid
- Avoid touching inside of container or lid
- Transport within 1 hour or refrigerate if delayed
C. 24-Hour Urine Collection
Purpose:
Evaluate kidney function, protein levels, creatinine clearance, hormone levels.
Method:
- Discard first morning urine, then collect all urine for 24 hours, including the last morning sample
- Store in large clean container, sometimes with preservative
- Keep container cool (refrigerated or on ice) during collection
- Label start and end time
D. Catheterized Urine Collection
Purpose:
For patients unable to void; ensures sterile collection.
Method:
- Use a sterile syringe to withdraw from catheter port (never from urine bag)
- Use aseptic technique
- Transfer into sterile container
E. Specialized Tests (e.g., pregnancy test, protein, glucose, ketones)
| Test | Sample Type | Notes |
|---|---|---|
| Pregnancy (hCG) | Early morning urine | Most concentrated |
| Glucose/Ketone | Random or fasting urine | Benedict’s or dipstick test |
| Protein | 24-hour or random | Dipstick or sulfosalicylic acid test |
II. FAECES COLLECTION METHODS FOR VARIOUS TESTS
A. Routine Stool Examination
Purpose:
Check for color, consistency, blood, mucus, pus, undigested food, etc.
Method:
- Container: Clean, dry, wide-mouthed container with tight lid
- Sample: 5–10 grams (about a teaspoon)
- Avoid contamination with urine or water
- Collect from freshly passed stool, preferably morning sample
B. Stool for Ova, Parasites, and Cysts (O&P Test)
Purpose:
Detect intestinal parasites (e.g., Giardia, Ascaris, Entamoeba histolytica)
Method:
- Fresh stool collected in sterile container
- Often requires 3 samples on alternate days
- No urine or water contamination
- Sample should reach lab within 30 minutes for trophozoite detection
- For preserved samples, formalin or PVA preservatives may be used
C. Stool for Occult Blood (FOBT)
Purpose:
Screen for hidden blood in GI tract (polyps, cancer, ulcers)
Method:
- Small sample from 2–3 different areas of stool
- Avoid red meat, NSAIDs, vitamin C 2–3 days before the test
- Use test kit or cards provided by the lab
- Usually 3 samples over 3 days
D. Stool Culture and Sensitivity
Purpose:
Detect bacterial infections (Salmonella, Shigella, E. coli, etc.)
Method:
- Fresh stool in sterile container
- Transport to lab within 30–60 minutes
- May require special media or transport systems
E. Stool for Reducing Substances (e.g., Benedict’s Test)
Purpose:
Diagnose carbohydrate malabsorption (e.g., lactose intolerance in infants)
Method:
- Fresh stool sample
- Test should be done immediately or refrigerated promptly
F. Stool for Fat (Steatorrhea Test)
Purpose:
Detect fat malabsorption in disorders like celiac disease, chronic pancreatitis
Method:
- Collect 72-hour stool (in some cases) or a single large sample
- Use Sudan III stain in lab for fat globules
3. GENERAL NURSING/CLINICAL GUIDELINES
For All Samples:
- Educate patient on proper collection technique
- Ensure sample is not contaminated
- Label container with:
- Patient’s name
- Date & time
- Type of test
- Use standard precautions (gloves, hand hygiene)
- Send samples to lab promptly (or store as required)