ENGLISH-msn-1-unit-5-Fluid and electrolyte-part-4 :-UPLOAD
Explain/Define of hypokalemia:
- Potassium is the major intracellular cation. About 98% of the body’s potassium is found inside cells. Potassium enters cells during the formation of new tissue and during the conversion of glucose to glycogen. When tissue breakdown occurs, potassium leaves the cells, and this can occur mainly due to trauma, dehydration, or starvation. Potassium acts as an important electrolyte in the conduction of nerve impulses and the contraction of skeletal and smooth muscles.
- Normal potassium levels are 3.5 to 5.5 meq/liter.
Hypokalemia is a metabolic condition in which In which the concentration of potassium is lower than the normal potassium concentration, it is called hypokalemia. - In the condition of hypokalemia, the potassium level is less than 3.5 meq/liter.
- ( In hypokalemia potassium level is less than < 3.5 meq/liter)
Explain Etiology/cause of Hypokalemia (Cause of Hypokalemia) ):
- Due to low intake of potassium in the diet.
- Due to high loss of potassium through the kidneys.
- Due to increased activity of aldosterone.
- Vomiting,
- Prolonged gastro intestinal tract Saxon
- Due to excessive sweating.
- Due to excess fluid loss through the gastrointestinal tract.
- Due to loss of potassium from the body when taking diuretic medicine.
- Due to any trauma and fluid loss.
- Due to a tumor in the adrenal gland.
- Eating Disorders.
- Diabetic Ketoacidosis.
- Treatment of Acidosis,
- Metabolic Alkalosis,
- Aldosteronism or Cushing’s Syndrome.
- Renal Tubular Acidosis,
- Medication: potassium losing diuretic, digoxin and corticosteroids.
- Antibiotics: amphotericine B, carbenicillin and gentamicine.
Explain the clinical manifestation/sign and symptoms of the Hypokalemia :
- Dizziness,
- Abnormal heart rhythm.
- Fatigue.
- Lethargy (low energy).
- Confusion.
- Nausea and vomiting.
- Orthostatic hypotension (sudden dizziness when sitting or lying down).
- Cardiac arrest.
- Hyporeflexia,
- Parasthesia (numbness and tingling),
- Loss of appetite.
- Constipation.
- Muscle weakness.
- Pain.
- Polyuria
- Nocturia,
- Excessive thirst,
- Respiratory distress,
- Paralysis,
- (Rhabdomyolysis: breakdown of muscle tissue).
Explain the Diagnostic evaluation of the Hypokalemia:
- History taking,
- Physical examination,
- serum pottasium <3.5meq/liter.
- Metabolic alkalosis.
- 24 hour urin potassium excretion test.
- Ecg changes.
- test for kidney function ( BUN and creatinine), glucose, Magnesium, And phosphorous.
- Medical Management of the Hypolkalemia:
- If there is mild to moderate level of hypokalemia, then it can be removed by oral potassium supplement.
- If the patient has severe potassium deficiency, then he has to take potassium intravenously. Daily 40 to 80 meq/day.
- Ask the patient to take potassium rich diet orally.
Ex: Berries, bananas, strawberries, melon, organ milk, chicken, beans, broccoli, carrots, potatoes, raisins, raisins, dried grapes, all these foods are rich in potassium. - If the patient cannot take it orally, then slowly infuse him with intravenous potassium chloride (KCL).
- Check whether the patient is producing adequate amounts of urine.
- Check whether the patient has signs and symptoms of hypokalemia.
- Monitor ECG.
- Generally, the patient needs a low sodium diet and a high protein diet.
- Routine Dietary Potassium Intake An average adult should consume 50 to 100 meq/day.
Nursing Management of Hypokalemia:
- Take a proper health history and health assessment of the patient to find the cause of the condition of hypokalemia.
- Administer potassium replacement therapy to the patient.
- Provide oral potassium by diluting it in proper water or juice.
- Check the IV site properly.
- Assess the patient’s intake-output chart every hour.
- Check the patient’s vital signs.
- Check the patient’s heart rate and rhythm.
- Assess the patient for toxicity of any digitalis group medicine.
- Check the patient’s potassium level when the patient is taking a diuretic medicine.
- Check the patient’s vital signs and pulse.
- Ask the patient to eat potassium-rich foods.
- When administering potassium intravenously, maintain proper infusion rates (not faster than 10-20meq/liter).
- Monitor the patient’s muscle tone and strength.
- Monitor the patient’s bowel sounds.
- Maintain the patient’s intake-output chart.
- Ask the patient to consume adequate amounts of potassium in the diet to prevent potassium loss.
Explain /Define of hyperkalemia:
- Hyperkalemia is a condition in which the potassium level in the body or blood is increased above the normal potassium level.
- Normal potassium level is 3.5 to 5.5 meq/liter .
- In hyperkalemia the potassium level is greater than 5.5meq/liter.
Etiology/causes of Hyperkalemia:
- Due to excessive use of potassium-containing salts.
- If the intake of potassium in the body is high and the kidneys cannot excrete that amount of potassium from the body. Due to excessive use of oral or intravenous potassium supplementation.
- Acute renal failure.
- Chronic renal failure.
- Adrenal gland insufficiency.
- Glomerulonephritis: Infection and inflammation of the glomerulus, the filtering part of the kidney, is called glomerulonephritis.
- Metabolic acidosis.
- Rejection of kidney transplant.
- Lack of aldosterone.
- Addison’s disease.
- Type 1 diabetes.
- Burns.
- Hemolytic conditions.
- Rhabdomyolysis
- (breakdown of muscle tissue) from drugs, alcoholism, coma, or certain infection.
- Certain medications such as:
- ACE inhibitor,
- Potassium chloride,
- Heparin,
- Captopril,
- NSAID,
- Use of potassium sparing diuretic (ex:spironolactone).
Clinical manifestation/ sign and symptoms of Hyperkalemia.
- Irregular heartbeat,
- Low blood pressure,
- Chest pain,
- Arrhythmias,
- Abdominal pain,
- Nausea,
- Vomiting,
- Palpitation,
- Muscle twitching and cramps.
- Diarrhea.
- Cardiac arrhythmia.
- Ecg changes.
- Muscular weakness and paralysis.
- Confusion and coma.
- Ventricular dysrhythmia and cardiac arrest.
- Respiratory failure.
- Tingling and numbness sensation.
- Cardiac arrest.
- Anemia.
- Muscle weakness.
Diagnostic evaluation of Hyperkalemia.
- History taking,
- Physical examination,
- Serum potassium levels are (greater than) > 5.5 meq.
- Metabolic acidosis-serum pH falls below 7.35.
- Ecg changes:
- Elevated T waves,
- Widened QRS complex,
- Prolonged PR interval,
- Flattened or absent P waves,
- Depressed ST segment.
Medical management of Hyperkalemia:
- Treatment of hyperkalemia depends on the cause, severity of hyperkalemia, symptoms, and overall health of the patient.
- The patient should consume a low-potassium diet.
- Continuous cardiac monitoring of the patient should be done.
- The patient should be provided with proper intravenous fluids.
- Continuous ECG monitoring of the patient should be done.
- Medications that reduce the amount of potassium in the body should be provided to the patient.
- If the patient has severe hyperkalemia, dialysis is also necessary.
- If the amount of potassium does not decrease through medication, calcium gluconate should be administered intravenously to the patient.
- The patient should be provided with sodium bicarbonate so that potassium can shift into the cells for a while.
- The patient’s serum potassium level should be checked continuously.
- Medications that increase the amount of potassium in the body should be discontinued.
- The patient should be administered 25% or 50% glucose and insulin intravenously to allow potassium to reenter the cell from the extracellular space.
- To reduce the effects of hyperkalemia, calcium should be provided intravenously for a short time to protect the heart and muscles.
- The patient should be provided with diuretic medication.
Nursing care for hyperkalemia (nursing management of hyperkalemia):
- Assess the patient’s intake output chart.
- The patient should be given potassium by oral or parenteral route.
- The patient should be advised to avoid potassium-rich foods, such as fruit juice.
- Assess the patient’s pain level and provide comfort measures.
- Assess the patient’s vital signs.
- Check the patient’s heart rhythm.
Calcium imbalance:

- Calcium is the most abundant mineral in the human body. It is critical and good for health.
- The normal calcium level in the body is 9 to 11mg/dl or 4.5 to 5.5 meq/liter.
- Calcium is a mineral that is mainly found in bones and teeth. More than 99% of calcium is found in bones. Calcium is an important component of bones and teeth. Calcium is important in nerve impulses and contraction and relaxation of muscles. Plays a role and includes cardiac muscles. Calcium plays an important role in blood clotting, muscle and nerve function.
Explain/define hypocalcemia:
- When there is not an adequate amount of calcium in the body and blood, it is called hypocalcemia. When the calcium level in the body is less than the normal level, it is called hypocalcemia.
- Normal calcium level is 4.5 to 5.5 meq/liter or 9 to 11mg/dl.
- (In hypocalcemia the level of calcium in blood is less than 9 mg/dl or 4.5 meq/ liter. )
Etiology/cause of Hypocalcemia (Causes of Hypocalcemia):
- The thyroid gland is underactive due to any disease or damage due to surgery.
- Due to excessive binding of calcium iron.
- Large amount of citrate in the blood.
- Hypoalbuminemia (low albumin level).
- Alkalosis.
- Hyperphosphatemia (high amount of phosphate).
- Due to insufficient calcium intake in the diet.
- Due to inadequate vitamin D intake in the diet.
- Due to chronic renal failure.
- Due to inadequate absorption of calcium in the gastrointestinal tract.
- Intestinal fistula. (Fistula is an abnormal connection between two organs).
- Crohn’s disease,
- Chronic inflammatory bowel disease.
- Due to deficiency of parathyroid hormone and vitamin D.
- Severe burns or infections. Osteoporosis (this is a bone condition in which the bones are weak and fragile, which can easily break down).
- Due to infection of the pancreas.
- Kidney failure.
- Due to low amount of magnesium in the blood.
- Certain types of drugs such as diuretics, estrogen replacement therapy, glucose, calcium channel blockers, insulin and magnesium are responsible for causing the condition of hypocalcemia.
- Biphosphate therapy.
- Certain types of leukemia (blood cancer) and blood disorders.
- Toxic shock syndrome.
Clinical manifestation/ sign and symptoms of Hypocalcemia (Tell the symptoms and signs of hypocalcemia).
- Osteoporosis (Bone becomes weak and fragile which can easily break down due to calcium deficiency).
- Anxiety and irritability,
- Pathogenic fracture (Bone becomes weak due to any kind of disease and hence it can easily break down).
- Tetany (Tetany: Muscle spasm occurs due to calcium deficiency and parathyroid gland being less active).
- Tingling sensation occurs around the nose and on the tips of the fingers.
- Tingling and numbness sensation also occurs in the hands and feet.
- Muscle spasm.
- Heart rate becomes irregular and increases.
- Nausea, vomiting.
- Blood pressure increases.
- Changes in mental status.
- Deep tendon reflexes become hyperactive.
- Gastrointestinal tract becomes enlarged.
- GI tract becomes enlarged, causing diarrhea and abdominal cramps.
- Cardiac arrhythmias.
- Laryngospasm (spasm of the vocal cords, causing difficulty in speech).
- Hypotension.
- Dry skin.
- Dermatitis (inflammation of the skin).
- Skin hyperpigmentation.
Chvostek sign:
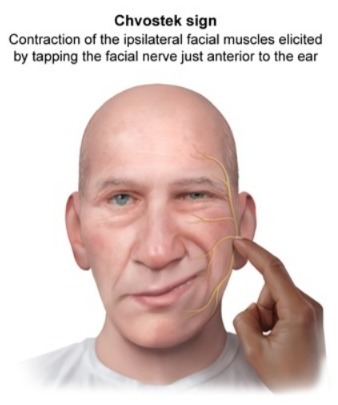
- Chvostek sign is a clinical sign that occurs during hypocalcemia and indicates an increase in neuromuscular excitability.
- When the facial nerve is excited by tapping the front of the ear, the facial muscles, especially the eye, nose, and lip, contract. There is an involuntary contraction or twitching around the (lip).
- This reaction is called a positive Chvostek sign. It is a major sign of hypocalcemia, which is caused by a decrease in the calcium ion concentration in the body.
Trousseau sign:
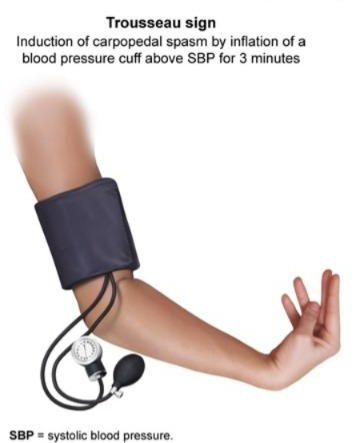
- Trousseau sign is a clinical sign that occurs in the presence of latent tetany or hypocalcemia.
- When a blood pressure cuff is placed on a patient’s arm and the systolic pressure is measured, When the blood pressure cuff is inflated to a pressure slightly higher than the systolic pressure for 2 to 3 minutes, carpal spasm, i.e. involuntary contraction of the fingers and wrist, occurs.
- This reaction is called a positive Trousseau sign, which is indicative of hypocalcemia.
Diagnostic evaluation of the Hypocalcemia (Diagnostic Evaluation of Hypocalcemia):
- History Taking,
- Physical Examination.
- Serum Sodium Level is Less than <8.5 mg/dl.
- Low Platelet Count.
- Increase Parathyroid Hormone Level.
- Ecg shows lengthened Qt interval, Prolong St segment, arrhythmias.
- Serum protein levels also change because serum calcium binds to albumin to a small extent.
Medical management of Hypocalcemia:
- Provide calcium supplementation to patients with calcium deficiency.
- Provide vitamin D in proper amounts.
- Provide calcium gluconate or calcium chloride intravenously to patients with acute or severe hypocalcemia.
- Calcium supplements should be given one to two hours after meals to increase intestinal absorption.
- If the patient has severe hypocalcemia with cardiac arrhythmia and tetany, provide calcium salts immediately.
- Provide calcium carbonate in the initial stage of hypocalcemia.
- Provide calcium carbonate at 250 or 500 mg/tablet.
- Provide calcium gluconate and calcium chloride to the patient.
- If there is a serious condition, provide calcium chloride to the patient.
- Provide calcium carbonate in the initial stage of hypocalcemia.
- Take vitamin D rich foods such as milk or dairy products to ensure proper absorption of calcium.
Nursing Management of Hypocalcemia:
- Check the patient’s vital signs and blood pressure.
- Administer calcium intravenously to the patient.
- Monitor the patient’s airway and respiratory status.
- Provide education to the patient and his family members about the sources of calcium.
- If the patient has undergone thyroid or neck surgery, monitor the patient carefully for signs and symptoms of hypocalcemia.
- Encourage the patient to exercise appropriately.
- Provide the patient with a quiet and comfortable environment to work in.
- Keep the patient’s bed at a low level and provide side rails.
- Encourage the patient to avoid caffeine intake.
- Smoking reduces the amount of calcium in the body, so encourage the patient to avoid smoking.
- Provide the patient with proper calcium-rich diet education.
Hypercalcemia:
- Hypercalcemia is a condition in which the level of calcium in the blood and body increases above 11 milligrams/deciliter (11mg/dl) or 5.5 meq/liter (5.5 meq/liter).
- Normal serum calcium level is (9 to 11 mg/dl or 4. 5 to 5.5meq/liter).
- Hypercalcemia The level of calcium is greater than > 11mg/dl or 5.5 meq/liter)
Etiology/cause of Hypercalcemia:
- Overactive parathyroid gland.
- Excessive release of calcium from bones due to bone cancer.
- Dehydration, loss of water from the body.
- Multiple myeloma,
- Multiple fractures,
- Immobilization,
- Intake of high amounts of calcium in the diet.
- Prolonged bed rest.
- Excessive absorption of calcium due to high amounts of vitamin D.
- Tumors that destroy bones.
- Excessive intake of calcium and vitamin D.
- Excessive consumption of milk products.
- Chronic kidney failure.
- Alkalosis.
- Excessive dietary intake of calcium.
- Due to high amounts of calcium absorption due to vitamin D.
- Chronic kidney disease.
- Due to use of certain medications such as thiazide diuretics.
- Inherited kidney or metabolic conditions.
Clinical manifestation/sign and symptoms of hypercalcemia:
- Increased heart rate.
- Increased blood pressure.
- Muscle weakness.
- Loss of appetite.
- Bone pain and pathological fractures.
- Constipation.
- Vomiting.
- Abdominal pain.
- Body ache.
- Decreased blood clotting capacity.
- Excessive urination (polyuria) and excessive thirst (polydipsia).
- Fatigue (lethargy).
- Formation of stones in the kidneys and buildup of waste products in the body.
- Confusion.
- Heart block.
- Difficulty speaking.
- Excessive sleepiness.
- Headache.
- Irritability.
- Depression.
- Memory impairment.
- Mood swings.
- Confusion.
- Renal stones.
- Decreased deep tendon reflex.
- Coma.
Diagnostic evaluation of the Hypercalcemia:
- History taking
- Physical examination.
- Serum calcium level is greater than 11 mg/dl.
- ECG shows sign of heart block. Shortened QT interval and ST segment.
- X-ray may reveal the presence of osteoporosis, bone cavitation or urinary calculi.
- Urinalysis.
- Decreased parathyroid hormone.
- Sulkovich urine test shows increased calcium precipitation.
Medical management of the Hypercalcemia:
- If the patient has a condition of severe hypercalcemia, then he needs to be hospitalized immediately.
- Provide normal saline to the patient who has a condition of severe or acute hypercalcemia and maintain his hydration status.
- Provide normal saline to the patient who has nausea, vomiting, polyuria.
- Provide Lasix (Frusemide) to the patient who is taking diuretic medicine so that the amount of calcium is excreted from the body.
- Use medication that binds calcium and releases it from the body.
- Ask the patient to undergo embolization.
- Advise the patient to drink plenty of fluids.
- Provide reassurance to the patient and his family members.
- Calcitonin helps to lower the serum calcium level.
- Biphosphate helps to increase the amount of calcium absorbed into the bones.
- Administer glucocorticoids.
- Dialysis.
Nursing management of the Hypercalcemia:
- Maintain the patient’s intake output chart.
- Ask the patient to drink adequate amounts of fluids.
- Advise the patient to eat a low calcium diet.
- Provide reassurance to the patient’s parents and family members.
- If the patient’s serum calcium level increases above 5.5 meq/liter, check the patient for cardiac arrhythmias.
- If the patient is using diuretics or normal saline, check the patient for signs and symptoms of heart failure.
- If the patient is receiving glycosides, check for toxicity such as loss of appetite, nausea, vomiting, bradycardia, etc.
- If the patient’s bones are weak, carefully change the patient’s position.
- If the patient is bedridden, change the patient’s position frequently and ask the patient to do range of motion exercises.
- Check the patient’s vital signs.
- Assess the patient’s respiratory status.
- Check the patient’s heart sound.
- Ask the patient to do physical activity to maintain weight.
- Ask the patient to eat adequate amounts of fiber-rich food to relieve constipation.
- Ask the patient to take a low calcium diet and increase fluid intake.
- Ask the patient to do activity so that calcium can be reduced from the body.
Introduction of Magnesium:

- Magnesium is an essential component that regulates over 300 enzymes that are responsible for many body functions.
- Magnesium works as a cofactor for many body enzymes.
- Magnesium plays an important role in the body’s metabolic activity.
- Magnesium is essential for the relaxation of smooth muscles, such as those around the bronchial tubes. muscles and skeletal muscle contraction and neurons that are located in the brain.
- Magnesium converts what is in the body into energy and builds up in proteins Helps in the formation of blood clots and maintains the level of calcium in the blood.
- Magnesium prevents Cardio Vascular Disease and prevents irregular heart beat.
- Magnesium helps in preventing heart attacks.
- Magnesium prevents stroke conditions.
- (The normal value for serum Magnesium is 1.5 to 2.5 meq/liter ) or (1.8-3.0mg/dl) If there is an imbalance in magnesium, it is called hypomagnesemia or hypermagnesemia.
Explain/ define Magnesium deficit (hypomagnesemia):
- Hypomagnesemia is an electrolyte disturbance in which there is an abnormally low level of magnesium in the blood, less than the normal magnesium level.
- ( Normal adult value of magnesium is 1.5-2.5meq/liter.)
- (In hypomagnesemia level of magnesium is less than <1.5 meq/liter)
Etiology/cause of Hypomagnesemia (causes of hypomagnesemia):
- Due to excessive loss of magnesium from the body.
- Due to low intake of magnesium.
- Due to protein calorie malnutrition.
- Due to lack of proper absorption in the intestine.
- Due to malnutrition or starvation.
- Due to excessive fluid loss from the body.
- Due to excessive urine excretion from the body.
- Severe diarrhea.
- Crohn’s disease.
- Due to excessive diuretic administration.
- Use of certain medications including amphotericin,
- Cisplatin, Aminoglycoside.
- Gastrointestinal fistula.
- Renal damage.
- Total parenteral nutrition.
- Excessive urination (polyuria).
- Alcoholism.
- Malabsorption.
- Due to high blood calcium levels in the body.
- Hyperaldosteronism.
Clinical manifestation/sign and symptoms of Hypomagnesemia:
- Positive Trousseau’s and Chovostek signs.
- Loss of appetite.
- Nausea.
- Vomiting.
- Weakness.
- Apathy.
- Muscle weakness.
- Parasthesia: (tingling or numbness sensation).
- Movement slow and involuntary.
- Irritability,
- Tetany,
- Leg and foot cramps.
- Staggering.
- Tremors,
- Ataxia: Lack of normal coordination.
- Carpopedal spasm (spasm of the muscles of the hand).
- Depression.
- Irritability.
- Psychotic behavior.
- Staggering.
- Vertical nystagmus: Involuntary movement of the eyeball.
- Cardiac dysrhythmias.
- Extreme agitation.
- Insomnia.
- Delirium.
- Auditory and visual hallucinations.
- Diagnostic evaluation of the Hypomagnesemia:
- History taking.
- Physical Examination.
- Serum Magnesium lower than 1.5 meq/liter.
- Nuclear Magnesium Resonance Spectroscopy.
Electrocardiogram shows:
- A) Prolonged PR and QT intervals,
- B) Widening QRS,
- c) ST segment depression.
- D) Flattened T waves.
- E) Prominent U wave.
Medical Management of Hypomagnesemia:
- Treatment of hypomagnesemia is provided based on the severity of the deficiency and the clinical effects.
- The aim of treatment is to identify and eliminate the cause of hypomagnesemia and replace the amount of magnesium in the body.
- Provide oral magnesium replacement to patients with mild symptoms.
- Provide intravenous magnesium replacement to patients with very severe signs and symptoms and advise patients with mild hypomagnesemia to eat a magnesium-rich diet.
- Like: Green leafy vegetables,
Nuts,
Legumes,
Whole grains,
Sea food, etc.
Take magnesium preparations such as magnesium oxide as they contain high amounts of magnesium. - Provide intravenous magnesium sulfate to patients with severe hypomagnesemia. (10 to 40 meq/liter).
- If the patient has conditions like cardiac arrhythmia,
Obstetrics problem,
Electrolyte disturbance,
Asthma.
etc., provide intravenous magnesium sulfate.
Nursing management of the Hypomagnesemia.
- Ask the patient to take magnesium rich food.
- Monitor the patient’s level of consciousness.
- Monitor the patient’s breathing pattern.
- Assess the patient for changes in E.C.G. Assess Ecg changes in a person who is taking digitalis group of medicines.
- Advise the patient to avoid intake of diuretics and laxatives in excessive amounts.
- Monitor the patient’s bowel sounds and abdominal distension.
- Check the patient’s reflexes before infusing magnesium.
- Provide the patient with a quiet, dark room.
- Provide the patient with a low bed and keep its side rails high to prevent falls if the patient is dizzy.
Explain/ define hypermagnesemia:
- Hypermagnesemia is an electrolyte imbalance in which the level of magnesium in the body increases to more than 2.5 meq/liter.
- A normal level of magnesium in the body is important for the function of the heart and nervous system.
- (In hypermagnesemia the level of magnesium is greater than > 2.5 meq/liter).
Etiology/cause of the Hypermagnesemia:
- Hemolysis,
- Renal insufficiency,
- Excessive magnesium intake due to excessive use of antacids and laxatives.
- Hypothyroidism.
- Lithium therapy.
- Diabetic ketoacidosis.
- Adrenal insufficiency
- Overdose of magnesium salts.
- Severe dehydration.
- Chronic Renal Insufficiency.
clinical manifestation / sign and symptoms of the Hypermagnesemia
- Nausea and Vomiting,
- Weakness,
- Difficulty breathing,
- Low blood pressure (hypotension),
- Bradypnea,
- Fatigue,
- Drowsiness,
- Skeletal muscles Weakness,
- Diminished reflexes,
- Facial paresthesia,
- Flaccid muscles paralysis,
- Hypercalcemia,
- Arrhythmia,
- Diminished deep tendon reflexes to occur.
- Bradycardia.
- Breathing loss.
- Bradycardia.
- Cardiac failure.
Diagnostic evaluation of the Hypermagnesemia (Diagnostic evaluation of the Hypermagnesemia) Evaluation):
- History taking,
- Physical examination,
- serum Magnesium level is greater than 2.5 meq/liter.
- Elevated potassium and calcium levels.
- ECG changes: prolonged PR interval ,
- Tall t waves,
- Prolonged QT interval and Widened QRS.
Medical Management of the Hypermagnesemia
- Instruct the patient to take in more fluids.
- Provide diuretic medicine to the patient.
- Infuse calcium gluconate to prevent magnesium toxicity.
- Provide diet for patients who cannot pass magnesium through urine, i.e., if their kidney function is impaired.
Nursing Management of the Hypermagnesemia.
- Carefully monitor the patient.
- Maintain the patient’s intake output chart.
- Tell the patient not to eat magnesium-rich foods.
- Monitor the patient for lethargy or drowsiness.
- Avoid over-the-counter drugs that contain magnesium.
- Check the patient’s vital signs.
- Assess the patient’s respiratory function.
- Monitor Ecg changes like:( prolonged PR, prolonged QRS, and prolonged QT.)
- Advise the patient to eat a low magnesium diet.
Explain phosphorus (phosphorus):

- Phosphorus is an intracellular anion.
- Phosphorus is essential for the normal functioning of cells.
- Phosphorus is found primarily in combination with calcium in the teeth and bones.
- Phosphorus is the primary anion of intracellular fluid.
- About 85% of phosphorus is found in bones and teeth. About 14% is found in soft tissues and less than 1% in the blood. It is found in the extracellular fluid (ECF).
- Phosphorus is responsible for a number of important functions.
- Phosphorus is involved in the formation of ATP (adenosine triphosphate) and also supports bones and teeth.
- Phosphorus is involved in the utilization of vitamin D, acid-base homeostasis, nerve, And plays an important role in muscle activity and in the metabolism of carbohydrates, proteins, and fats.
- Normal serum phosphorus level 2.5 to 4.5 mg/dl (0.8 -1.5 mol. /liter), Phosphorus imbalance due to hypophosphatemia (hypophosphatemia: phosphorus amount is less than normal amount) and hyperphosphatemia (hyperphosphatemia: phosphorus amount is more than normal amount) like condition may arise.
Definition of hypophosphatemia:
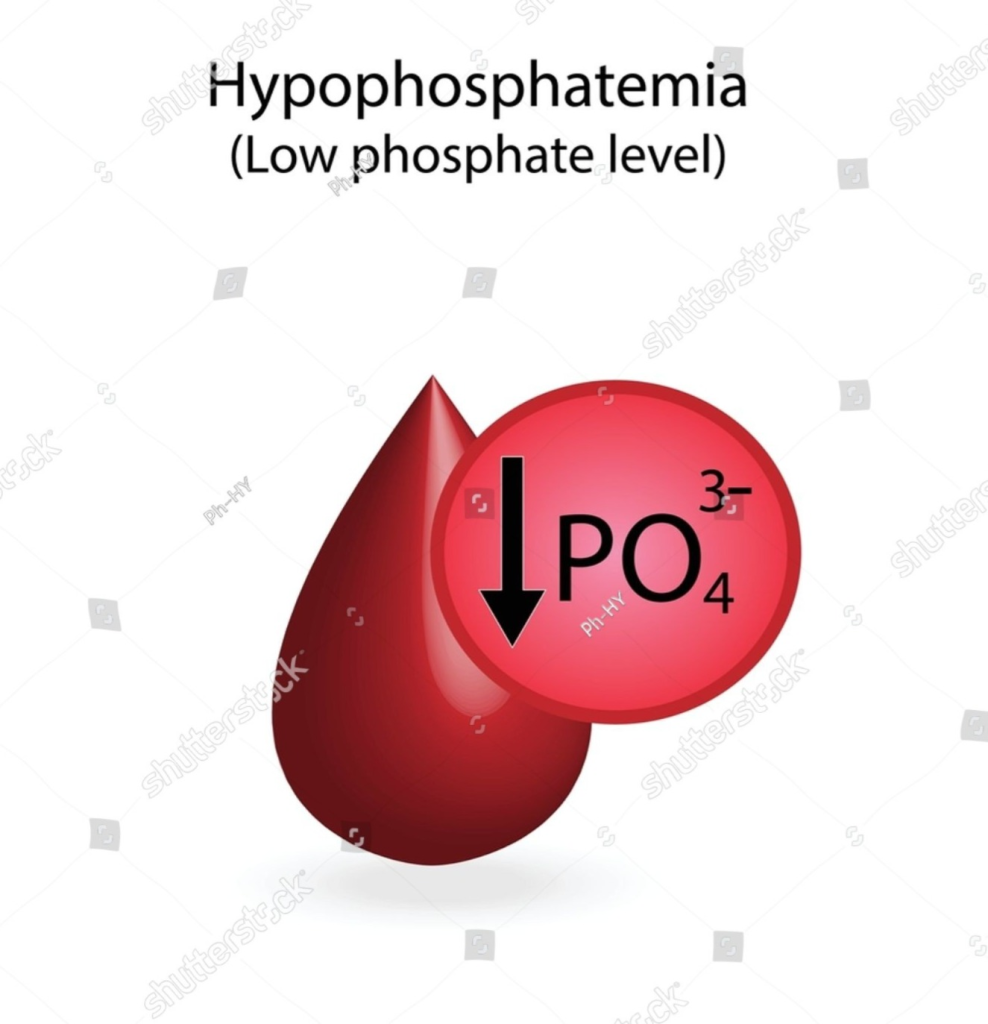
- Hypophosphatemia is a condition in which the level of phosphate in the body is lower than the normal level.
- Hypophosphatemia is caused by starvation and alcoholism.
- (normal Phosphate level is 2.5 to 4.5 mg/dl( 0.8 -1.5 mmol/liter))
- In mild hypophosphatemia ( 2-2.5mg/dl or 0.65 -0.81 mmol/liter),
- In moderate Hypophosphatemia (1-2mg/dl or 0.32 -0.65 mmol/ liter),
- In severe hypophosphatemia (<1mg/dl or 0.32 mmol/liter).
Etiology/ cause:
- Administer intravenous fluid without phosphorus.
- Hyperparathyroidism: This is a condition in which the parathyroid gland secretes too much parathyroid hormone.
A condition of hypophosphatemia may arise due to poor kidney function in which the kidney tubules are unable to reabsorb adequate amounts of phosphorus. - Recovery from certain diseases such as diabetic ketoacidosis or severe burns may cause a condition of hypophosphatemia.
- Due to excessive use of diuretic medicine.
- Due to malnutrition due to long-term alcoholism and not taking in adequate amounts of phosphorus.
- Due to any intestinal abnormality in which the intestine cannot absorb adequate amounts of phosphorus. Ex:chronic diarrhea.
clinical manifestation/(sign and symptoms):
- loss of appetite,
- weakness in skeletal and smooth muscles.
- tingling sensation sensation).
- Numbness.
- Paresthesia ( feeling of tingling and numbness sensation).
- Tremors.
- Bone pain.
- Weakness.
- Respiratory Respiratory insufficiency.
- Impaired neurologic function.
- Confusion.
- Memory loss.
- Stunned.
- Coma.
- Hemolytic Anaemia.
- Impaired leukocytes and platelet count.
Diagnostic Evaluation Evaluation):
- History tacking
- Physical examination.
- serum Phosphate level is less than 2.5 mg/dl.
- urin Phosphate level greater than 1.3 g/24 hours.
Medical management:
- Advise the patient to take phosphate in the diet.
- If phosphate replacement through diet is not possible, then provide medication supplementation to the patient.
- If medication is not curable, then provide phosphate intravenously to the patient.
- Advise the patient to take phosphate rich food.
Ex: green leafy vegetable 🥦,
peas (peas),
beans(🫘),
nuts( almonds),
chocalate,
beef liver,Etc.
Ask to take phosphate rich food. - If the patient has severe hypophosphatemia, then provide intravenous phosphate.
- the dose of Kpo4 2.5 mg/dl every 6 hourly.
- If the patient has severe hypophosphatemia, then provide intravenous phosphate.
- If the patient has severe hypophosphatemia, then provide intravenous phosphate.
- If there is hyperparathyroidism, the parathyroid gland should be surgically removed.
Nursing management:
- Check the patient’s serum electrolyte level.
- Instruct the patient to include phosphate-rich foods in their diet.
- Check the patient for muscle weakness.
- Check the patient’s mental status.
- Check the patient’s vital signs.
- Maintain the patient’s intake-output chart.
Defination of hyperphosphatemia (Hyperphosphatemia):

- Hyperphosphatemia is a condition in which there is an excess of phosphate in the body and blood. Phosphate levels increase above normal levels. Hyperphosphatemia is caused by excessive absorption of phosphate, decreased loss from the body, and excessive production. One of the main causes of hyperphosphatemia is renal failure.
Etiology/cause:
- Renal failure,
- Chemotherapy,
- Hypothyroidism (low thyroid levels) Due to),
- Respiratory acidosis,
- Diabetic ketoacidosis,
- Due to excessive intake of phosphate.
- Profound muscles necrosis.
- Hyperphosphatemia is a condition caused by excessive use of laxatives and enemas containing phosphate. It is caused by:
- Kidney failure causes phosphate to be excreted from the body and magnesium deficiency.
- Hypoparathyroidism is a condition in which the parathyroid gland does not produce adequate amounts of parathyroid hormone.
- Due to the inability of the kidneys to respond to parathyroid hormone, phosphate is not excreted from the body and the amount of phosphate in the body increases. is.
- The condition of hypocalcemia also increases the amount of phosphate in the body.
clinical manifestation/sign and symptoms :
- Tetany,
- Tingling sensation in the fingertips.
- Loss of appetite.
- Nausea.
- Vomiting,
- Muscle weakness weakness).
- Hyperreflexia
- Tachycardia.
- Decrease urine output.
- Visual disturbances.
- Palpitations.
- Depression Depression).
- Loss of memory.
- Convulsions.
- Calcification of soft tissue.
- Arteriosclerosis.
- Heart attack and stroke due to arteriosclerosis.
Diagnostic evaluation:
- History taking and physical examination.
- Serum phosphate level is greater than 4.5 mg/dl.
- Serum calcium level is less than 9 mg/dl. (serum calcium level is less than 9mg/dl).
- X Ray ,
- Provide the patient with vitamin D preparation to reduce phosphate levels.
- Use oral phosphate binders that can absorb phosphorus from the G.I track.
- (Provide lanthanum carbonate to dialysis patients). lanthanum carbonate provides to the dialysis patients.
- Use calcium salts such as (calcium carbonate, gluconate and chloride) which bind phosphate from the blood stream and reduce the level of phosphate in the blood.
- Use albumin salts such as albumin hydroxide which can bind phosphorus.
- If the patient has severe hyperphosphatemia, then calcium gluconate should be provided to the patient intravenously.
- Dialysis is an important treatment to remove excess phosphate from the blood.
- Foods rich in phosphate, such as dairy products, meat, nuts, and other high-protein foods, should be avoided.
Nursing Management:
- Assess the patient’s serum phosphate level.
- Check the patient for signs and symptoms of hypocalcemia.
- Properly monitor patients who are at high risk for hyperphosphatemia.
- Advise the patient to follow a low-phosphate diet.
- Instruct the patient to eat phosphorus-rich foods such as
- 🧀 Avoid phosphate-rich foods such as cheese, cream, nuts, whole grains, cereals, dried fruits, vegetables, sweetbreads, and milk foods.
- Once the patient’s phosphate level is normal, instruct the patient to avoid laxatives and enemas that are high in phosphate.
- Instruct the patient to check for any signs and symptoms of hypocalcemia and assess for any changes in the patient’s urine output.
- Advise the patient to eat a low-phosphate diet.
Chloride imbalance (Chloride Imbalance) :
Chloride is the major extracellular fluid anion and is found primarily in blood, intestinal and lymph fluids. Chloride is mainly found in gastric juice, pancreatic juice and sweat.
Define chloride deficit (Hypochloremia):
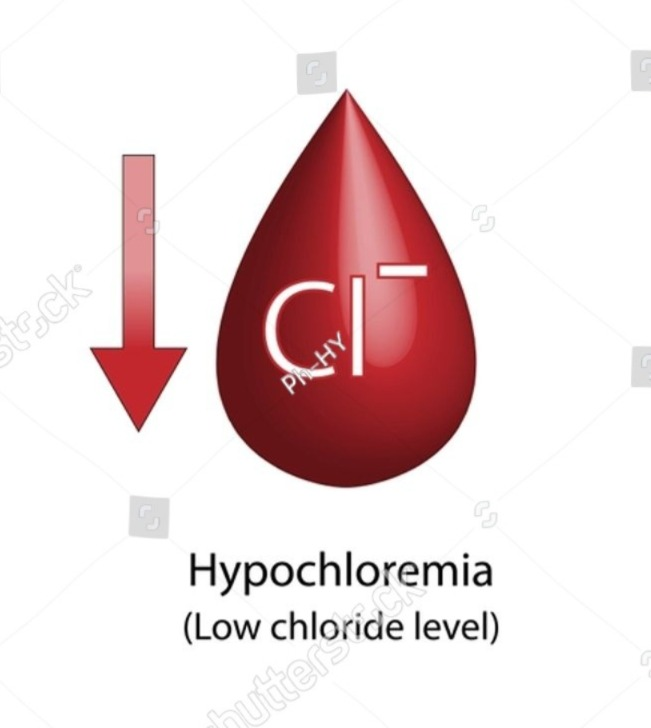
- The concentration of chloride depends on its intake.
- Excretion and reabsorption of chloride occur in the kidneys.
- Chloride is produced in the stomach as hydrochloric acid and a small amount of chloride is lost in the feces (stool).
- Hypochlorite is an electrolyte. There is an imbalance in which the chloride level in the body and blood is lower than the normal level.
- (Normal chloride level in adult is 97-107meq/liter. )
- (In hypocloremia the level of chloride is less than 97 meq/liter.)
- In hypocloremia if the chloride level is less than 97 meq/liter, it is called hypocloremia.
Cause/Etiology :
- Due to low intake of chloride.
- Due to low absorption of chloride.
- Due to low intake of sodium.
- Due to low intake of potassium.
- Due to prolonged use of diuretic medicine.
- Metabolic alkalosis.
- Due to excessive chloride release from the body due to diarrhea.
- Vomiting,
- Gastric suctioning.
- Gastric Surgery.
Signs and symptoms / clinical manifestation :
- Confusion,
- Apathy ( loss of interest),
- Disorientation.
- Excessive sleep.
- Staggering.
- Sitting in one place.
- Muscle wasting.
- Atrophy ( decrease in size of muscles and tissue).
- Hypotonia.
- Tetany.
- Deep tendon reflexes are hyperactive.
- Weakness.
- twitching.
- Muscle cramps.
- Cello breathing.
- Hyponatremia.
- Hypokalemia.
- Muscle cramps.
- Cardiac dysrrhythemias.
- Caesar (siezer).
Diagnostic evaluation:
- history taking and physical examination .
- serum chloride level is less than 97 meq/liter.
- serum ph is greater than 7.45.
- Serum co2 level is greater than 32 meq/liter.
- Serum Bicarbonate.
- Serum electrolyte.
- urine electrolyte.
- urin osmolarity.
Medical management:
- Ask the patient to take salty food orally.
- Inject the patient with 0.9% intravenously Administer sodium chloride.
- Administer ammonium chloride to the patient.
- Administer potassium chloride orally or intravenously to the patient.
- 10- 40 meq/liter potassium chloride administration.
- If potassium chloride is to be administered intravenously, it should not exceed 20 meq/liter.
- A uretic medicine that removes chloride from the body It should be avoided to give to the patient who excretes.
- Foods which have high amount of chloride should be given to the patient such as:
- Tomato juice (🍅 ),
- salty broth,
- canned vegetable,
- processed meat ,
- Provide food like fruits, etc.
- Bottled water excretes excess chloride from the body, so such water should be avoided.
- Ammonium chloride an acidifying agent may be used for metabolic alkalosis.
- The patient should have an increased chloride diet.
Nursing Management:
- Maintain the patient’s intake output chart.
- Assess the patient’s arterial blood gas values.
- Check the patient’s serum electrolyte levels.
- Check the patient’s level of consciousness.
- Check the patient’s muscle strength and momentum.
- If any changes are observed, report them to the physician immediately.
- Properly monitor the patient’s vital signs.
- Provide patient education or encourage a high-chloride diet.
Define hyperchloremia:
- Hyperchloremia is an electrolyte imbalance in which the level of chloride in the blood and body is below the normal value exceeds.
- ( Normal chloride level Is 97- 107 meq/liter).
- ( In hypercloremia the value of chloride is greater than108 meq/liter)
- Hypernatremia along with loss of bicarbonate from the body and metabolic acidosis increases the level of chloride.
Etiology/cause:
- Due to excessive chloride intake.
- Excessive chloride in the body Due to absorption.
- Hyperingestion of ammonium chloride.
- ureterointestinal anastomosis.
- Ingestion of excess amount of chloride through any route of the body like intra venously.,Orally,Nasofastric tube,Enema etc.
- Due to taking certain types of medicine like diuretics.
- Dehydration Because of.
- vomiting,
- diarrhea,
- sweating ,
- high fever,
- metabolic acidosis,
- Brain stem injury causes hyperventilation and hyperparathyroidism.
Clinical manifestation/ sign and symptoms :
- tachypnea (increase respiratory rate),
- weakness,
- fatigue,
- deep and rapid respiratory rate.
- Decreased thinking ability.
- Hypertension.
- Hypervolemia.
- Hypernatremia,
- Hyperchloremia,
- Decreased cardiac output.
- dysrrhythemias.
- Comma.
- Fluid retention.
Medical management :
- In severe hyperchloremic acidosis, treatment includes intravenous administration of sodium bicarbonate to increase bicarbonate levels, thereby increasing renal excretion of chloride ions.
- In mild hyperchloremia, administration of Ringer’s lactate solution, which allows the carbonate to enter the liver and treat the acidosis, is recommended. is.
- Administer diuretics to eliminate chloride.
- Sodium, fluid And keep chloride intake low or restricted.
- It is necessary to correct the cause of hyperchloremia and maintain electrolyte and acid-base balance.
Nursing Management:
- Monitor the patient’s vital signs.
- Check the patient’s arterial blood gas value.
- Assess the patient’s intake output.
- Assess the patient’s respiratory, neurologic, and cardiac systems. If there are any changes in them, immediately report them to the physician. To report.
- Educate the patient on diet.
- Acids and bases are formed as part of normal metabolic processes in the body.
- Acids are formed as end products of glucose, fat and protein and are fixed in the blood. It is called an acid because once it is formed, it does not undergo any changes.
- A weak acid, carbonic acid is formed when carbon dioxide and water combine in cellular metabolism.
- This acid is not fixed because it changes back to form bicarbonate and hydrogen.
Explain Acid Base Balance (Explain Acid Base Balance):
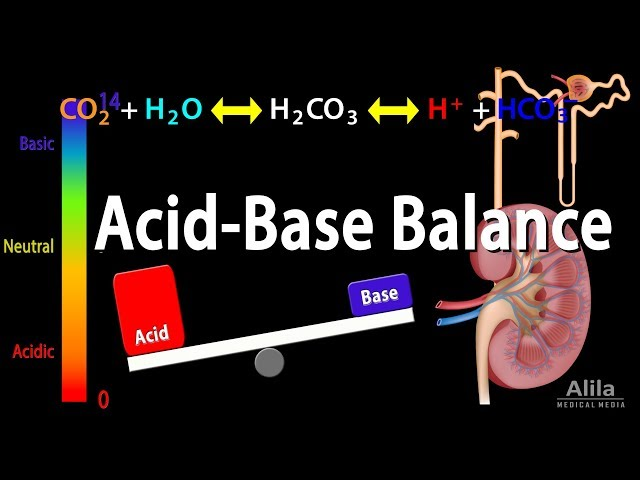
- Acids and bases are formed as part of normal metabolic processes in the body.
- Acids are formed as the end product of glucose, fat, and protein and are called fixed acids because once formed, they do not change.
- A weak acid, carbonic acid, is formed by the combination of carbon dioxide and water during cellular metabolism.
- This acid is a These acids are not fixed because they undergo changes back to form bicarbonate and hydrogen.
Explain Acid – base Imbalance. (Describe Acid Base Imbalance):
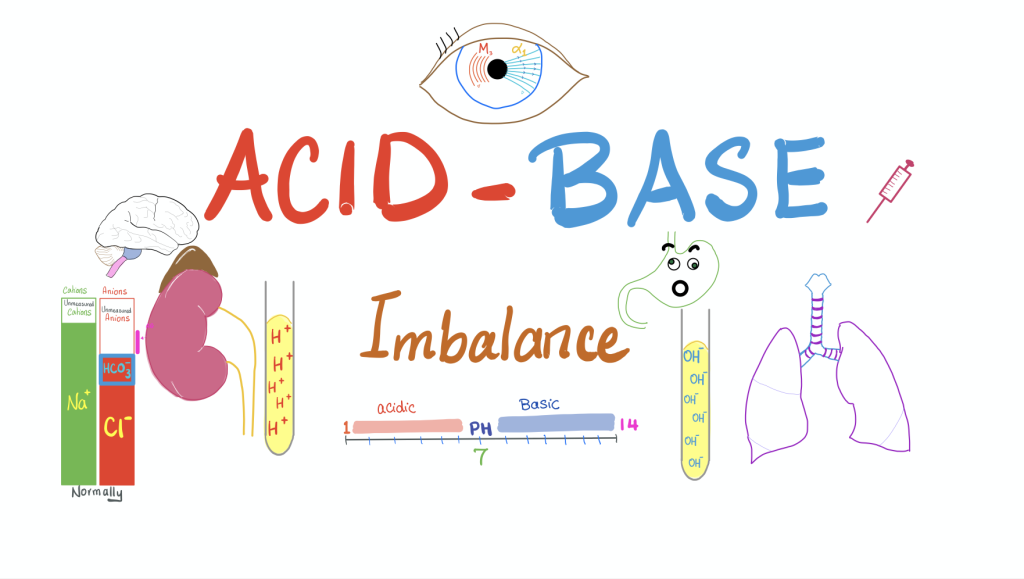
- Acids and bases are formed in the body as part of a normal metabolic process. is.
- To maintain acid and base balance, the kidneys secrete hydrogen ions (acid). Sodium reabsorbs iron and bicarbonate ions (base). Phosphate salts acidify and produce ammonium ions.
- Due to these, the normal blood pH is between 7.35 to 7.45 maintained in the body.
- Respiratory imbalance occurs when there is a problem with the oxygenation of the blood. Ex:hyperventilation,Stagnation of blood flow, etc .
- Metabolic imbalance occurs when kidney function breaks down.
- Fluid is injected ( ingestion) and lost (loss) and Due to other problems in the body organ.
- The acid base status of the patient can be obtained by taking the following samples from arterial blood.
- 1) ph (normal 7.35 to 7.45): measure hydrogen ion concentration.
- 2)PCO2 (normal 35 – 45 mmHg): partial pressure of carbon dioxide.
- 3)HCO₃⁻ Bicarbonate (normal 22 – 26 mEq/L)
- 4) PaO₂ (Partial Pressure of Oxygen Normal Value: 80 – 100 mmHg
- 5) O₂ Saturation : Normal Value: 95% – 100%

Describe respiratory acidosis (Respiratory acidosis):
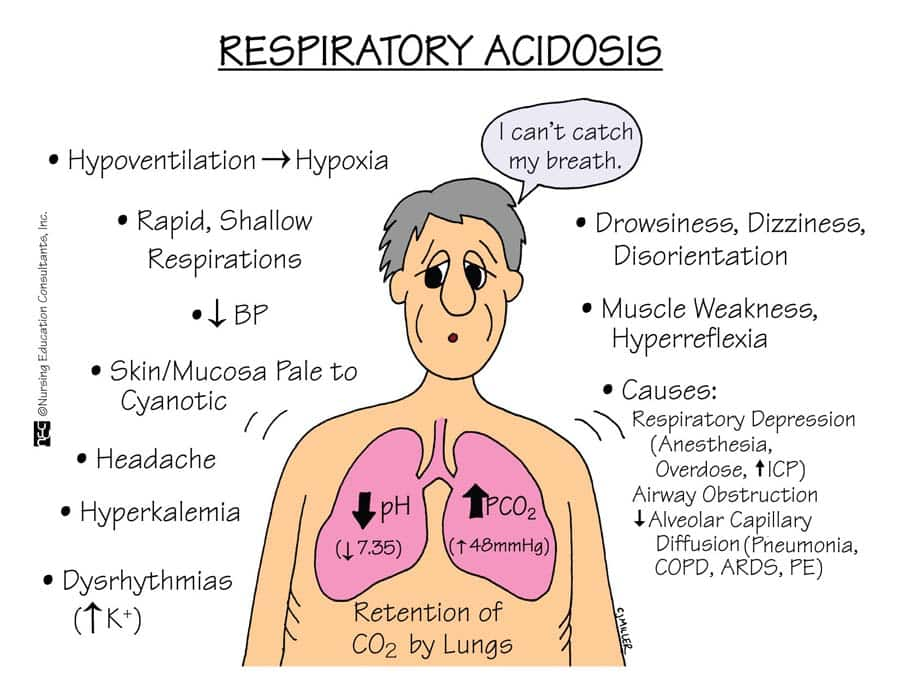
- Respiratory acidosis is a condition that occurs when the lungs are unable to remove all the carbon dioxide produced in the body from the body.
- Respiratory acidosis is a medical condition in which the ventilation of the body decreases ( hypoventilation) and the amount of carbon dioxide increases in the blood and due to this the blood pH decreases. This condition is called acidosis.
- Alveolar ventilation It decreases.
- In respiratory acidosis, the blood pH is less than 7.35.
- Pco2 is greater than 42 mmhg.
- This causes acid-base imbalance and due to this, the body fluid and especially blood It becomes acidic.
- Carbon dioxide is produced as the body’s cells repair and carbon dioxide accumulates. If sufficient carbon dioxide cannot be expelled from the body through alveolar ventilation, alveolar hypoventilation occurs. Due to this, the amount of paco2 in the body increases (hypercapnea).
Type of respiratory acidosis (type of respiratory acidosis)
- 1) Acute respiratory acidosis (acute respiratory acidosis)
- 2) chronic respiratory acidosis. ( Chronic Respiratory Acidosis )
1) Acute respiratory acidosis ( Acute Respiratory Acidosis ):
- Acute respiratory acidosis occurs when ventilation fails suddenly.
- In Acute respiratory acidosis paco2 is Elevated above the upper limit of reference range (over 47 mmhg )( ph <7.35) .
- Acute respiratory acidosis is caused by hypoventilation and is mainly due to disease of the central respiratory center located in the cerebral cavity or due to any drugs, myasthenia gravis, Guillain-Barré syndrome etc. or Airway obstruction due to COPD (chronic obstructive pulmonary disease) It can also be due to .
2) cronic respiratory acidosis ( Chronic Respiratory Acidosis):
- Chronic Respiratory Acidosis is a Pulmonary disease Or it may be due to a long-standing disease or secondary to any other disease.
- In chronic respiratory acidosis, pCO2 exceeds the normal range.
- Normal blood pH ( 7.35 to 7.45). Elevated serum Bicarbonate ( hco3- >30mm hg ).
- Chronic respiratory acidosis is caused by all disorders, including COPD ( copd : cronic obstruction Pulmonary disease).
- Chronic respiratory acidosis is caused by secondary obesity, hypoventilation syndrome, neuromuscular disorder , ventilatory defects and intestinal fibrosis, and thoracic It is seen due to deformity and also due to lung disease.
cause/ Etiology (cause)
- drugs : narcotics,
- Anaesthetic,
- Hypnotics,
- Sedative etc,
- Central Nervous System Ma trauma such as medullary injury that ventilatory drive Impaired does.
- Airway obstruction.
- parenchymal lung disease.
- Chestvolddisorder.
- severe kyphoscoliosis status post thoracoplasty.
- FLAIIL CHEST.
- alkalosis spondylitis.
- Chronic metabolic alkalosis which reduces alveolar ventilation.
- Neuromuscular diseases such as Gullin Barr syndrome, Poliomyelitis,
- Respiratory Hypoventilation occurs because the muscles cannot work properly.
- chronic obstruction Pulmonary disease ( copd),
- asthma,adult respiratory distress syndrome.
- chronic bronchitis.
- large pneumothorax.
- extensive pneumonia.
- Pulmonary Edema.
clinical manifestation/ sign and symptoms (symptoms and signs):
- difficulty breathing,
- extreme fatigue,
- chronic cough ,
- wheezing,
- confusion,
- irritability,
- lethargy,
- pulse rate increase,
- respiratory rate increase,
- blood pressure increase pressure,
- mental confusion,
- Felling of fullness in head,
- Anxiety,
- Delirium,
- Confusion,
- papilloedema,
- superficial blood vessels should be dilated.
Diagnostic evaluation:
- history taking and physical examination
- arterial blood gas analysis.
- pco2 greater than 45 mmhg.
- ph is below the normal limit 7.35 to 7.45.
- complete blood count tests.
- monitoring of serum electrolyte level.
- chest X Ray,
- Pulmonary function test,
- ct scanning,
- mri of brain,
- fluoroscopy,
- Ecg identify any cardiac involvement.
Medical management:
- The goal of treatment is to address the source of alveolar hypoventilation correct.
- If there is any disorder, correct it.
- provide bronchodilator medicine to patient.
- provide antibiotics medicine.
- administration supplementary oxygen therapy.
- Tell people to avoid smoking.
- Tell people to lose weight.
- provide Non invasive positive pressure ventilation.
- dialysis to eliminate toxic drugs.
- provide endotracheal intubation.
- tracheostomy.
- mechanical ventilation.
- provide antibiotic 💊 medicine.
- Administering the chesttube.
- PEEP : to prevent alveolar collapse.
- for Pulmonary emboli thrombolytics or anticoagulant therapy.
- Bronchoscopy to remove excessive secretion.
- Administration B agonist like ipratropium, bromide, methylxanthines.
- Provide oxygen therapy and corticosteroids to the patient.
Nursing Management:
- Monitor the patient’s arterial blood gas analysis.
- Assess the patient’s respiratory status.
- Provide the patient with a semi-folder position.
- Provide the patient with proper oxygen therapy.
- Listen to the patient’s breathing sounds. to ascultate.
- Respiratory alkalosis is a condition in which carbon dioxide levels are low due to excessive breathing and alveolar hyperventilation. In this, the partial pressure of carbon dioxide (pco2) is low (pco2 less than35 mmHg) and the blood pH is greater than 7.35.
Define respiratory alkalosis
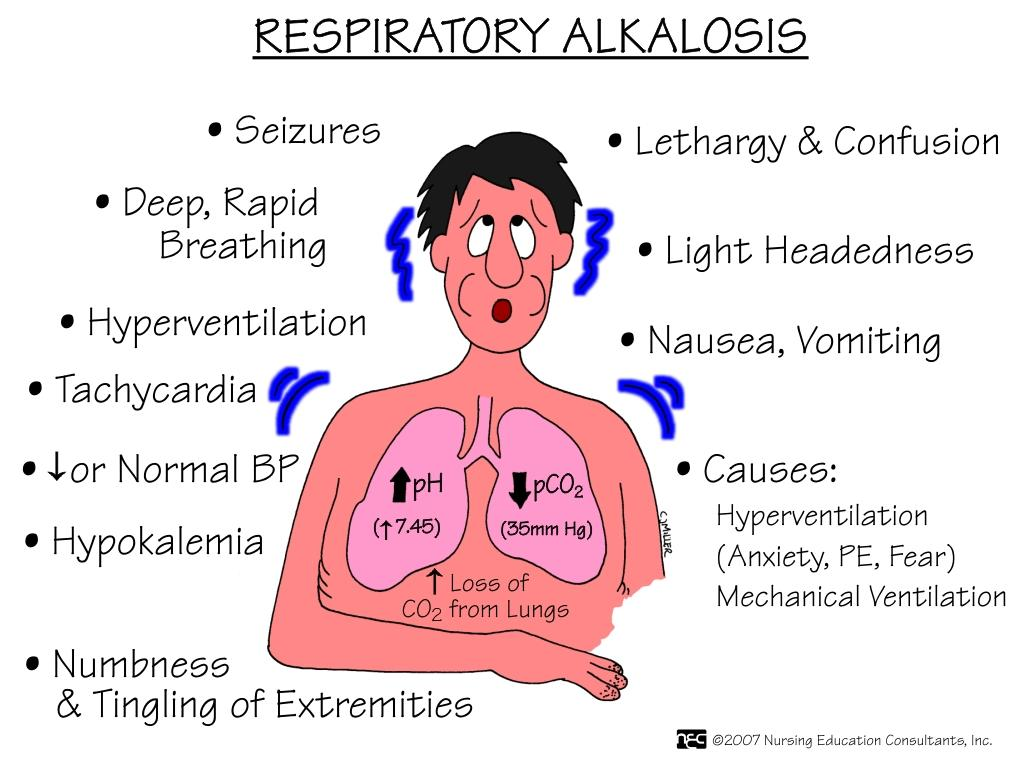
- Respiratory alkalosis is a condition in which carbon dioxide levels are low due to excessive breathing and alveolar hyperventilation. In this, the partial pressure of carbon dioxide (pco2) is low (pco2 less than35 mmHg) and the blood pH is greater than 7.35.
Etiology/ cause:
- Due to decreased PCO2 levels,
- Respiration rate increased.
- Lung disease.
- Mechanical ventilation.
- Pneumonia.
- Pneumothorax.
- In pulmonary ED.
- Pulmonary embolism.
- Emphysema.
- Chronic bronchitis.
- Acute asthma.
- Hyperthyroidism.
- Severe Anemia.
- Encephalitis.
- Hepatic failure.
- Hyperactive thyroid.
- Meningitis.
- Psychosis.
- Sepsis.
- Trauma.
- Tumor.
Clinical manifestation/ sign and symptoms :
- The signs and symptoms of respiratory alkalosis depend on its duration, its severity, and the extent to which the disease has progressed.
- Dizziness,
- Difficulty breathing,
- Numbness and tingling sensation,
- Numbness in the hands and feet Dizziness,
- Disorientation of time , place and person.
- Confusion.
- Chest pain.
- Trouble breathing.
- Low blood chlorine levels (hypochloremia).
- In the blood Low sodium levels (hyponatremia).
- Paresthesia (numbness and tingling sensation),
- Fainting.
- Syncope (a brief loss of consciousness when there is little or no blood flow to the brain).
- Tetania (muscle spasms).
- Breathing discomfort.
- Chest tightness.
- Tachypnea.
- Spasm in the muscles of the hand (Trousseau’s sign).
- Chovostek sign (When tapping on the front of the ear and on the cheek, facial muscles spasm occurs, this is seen in the body due to hypocalcemia).
diagnostic evaluation:
- History taking and physical examination.
- arterial blood gas level assessment.
- blood culture.
- brain MRI.
- Complete blood count.
- liver function test.
- serum analysis.
- sputum cultural.
- urin culture.
- chest X Ray.
- CT scan.
- lumber puncture.
- cytologic analysis.
- medical management (Medical Management):
- The management of respiratory alkalosis involves treating the cause of hyperventilation.
- To relieve hyperventilation, remove the toxic agent that has been ingested, such as salicylates,
Using gastric lavage,
Treatment of fever or sepsis,
Oxygen administration, - Asking the patient to do relaxation exercises can help reduce the patient’s anxiety level and improve breathing. is.
- The tidal volume and respiratory rate of a patient who is on mechanical ventilation are reduced.
- Do an arterial blood gas analysis of the patient.
- Give the patient sedative and antidepressant medicine.
- If the patient has respiratory alkalosis due to anxiety, then sedative and tranquilliser medicine helps the patient.
- If the patient is in a hyperadrenergic state, the patient should be provided with beta adrenergic blocker medicine.
nursing management:
- Check the patient’s respiratory rate.
- Check the patient’s vital signs.
- Ask the patient to breathe slowly.
- Provide reassurance to the patient.
- Relieve the patient’s anxiety.
- If the patient is hyperventilating, monitor the patient’s anxiety level properly and also check his respiratory level.
- If the patient’s anxiety level is very high, counsel the patient.
Explain metabolic acidosis:
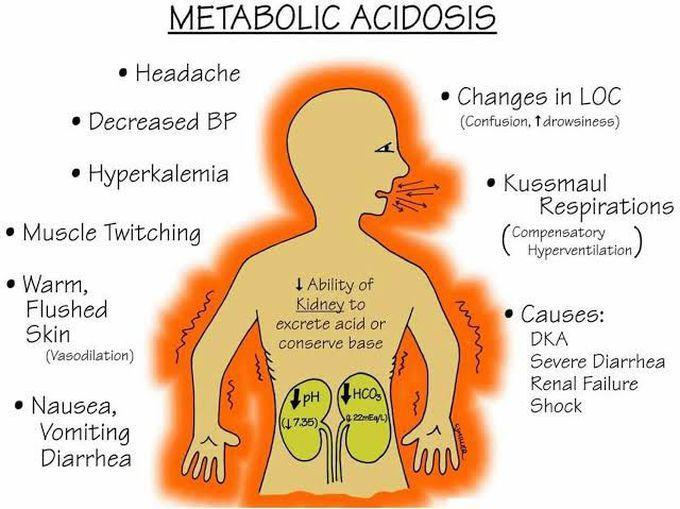
- Metabolic acidosis is an acid-base disorder in which too much acid is present in the body fluids because the kidneys cannot remove enough acid from the body and the concentration of bicarbonate in the body is low. Due to this, the condition of metabolic acidosis arises.
- The condition of metabolic acidosis arises when too much acid is produced in the body and the kidneys cannot remove enough acid from the body, due to which the condition of metabolic acidosis arises.
- Ex: blood pH is less than 7.35.
- Due to excessive production of hydrogen ions in the body and reduced production of bicarbonate (Hco3-).
Etiology/cause:
- Metabolic acidosis occurs when the body produces too much acid.
- When the kidneys do not remove adequate amounts of acid from the body.
- Lactic acidosis,
- Hyperchloremia Acidosis,
- Ketoacidosis,
- Kidney Disease (Distal Tubular Acidosis and Proximal Renal Tubular Acidosis),
- Chronic Renal Failure (Due to Accumulation of Sulfate, Phosphate and Urea),
- Poisoning by Aspirin, Glycol, or Methanol,
- Organism acid (salicylates ,thanol ,methanol, formaldehyde, glycol, paraldehyde and INH).
- Sulphate and metformin.
- Hypoaldosteronism.
- Lactic acidosis due to alcohol, cancer, liver failure, hypoglycemia and seizures.
- G.I.HCO3 loss due to diarrhea, fistula, ureterosigmoidostomy.
clinical manifestation sign and symptoms:
- fatigue,
- chest pain,
- palpitation,
- headache,
- Alteration of mental status,
- Severe anxiety,
- Loss of appetite,
- Impaired vision,
- nausea,
- vomiting,
- Stomach pain Abdominal pain,
- altered appetite,
- weight loss,
- muscular weakness,
- bone pain,
- deep and rapid breathing,
- increase the amount of carbon dioxide,
- fatigue,
- drowsiness,
- very sleepy,
- very tired,
- visual disturbance,
- retinal oedema,
Neurological :
- Lethargy (feeling tired),
- Stupar(stupid),
- Coma( Koma),
- Seizures,
- decrease cardiac output.
cardiac:
- Arrhythmias,
- Tachycardia,
- Hypotension (low blood pressure).
diagnostic evaluation:
- history taking and physical examination.
- arterial blood gas analysis to assess the severity of metabolic acidosis.
- blood pH is less than < 7.35.
- Decrese the Bicarbonate level <24 mmol/liter.
- Metabolic panel to check the cause and severity of metabolic acidosis.
- Complete blood count.
- ECG will detect dysrrhythemias caused by the increased ketoacidosis.
- Elevation of ketone indicate diabetic, alcoholism, and starvation.
- urin pH is less than 4.5.
- serum potassium greater than 5.5 meq/liter ang glucose level is greater than 150 mg/dl support the diagnosis of metabolic acidosis.
Medical Management:
- Provide sodium bicarbonate to the patient 50 – 100 mmol increases the acidity of the patient’s blood and neutralizes the acidity in patients whose blood pH is less than 7.20.
- If the metabolic acidosis is very serious, then the patient should undergo dialysis.
- If the patient has hypokalemia, i.e. low amount of potassium in the body, then the patient’s potassium level should be continuously monitored and measures should be taken to normalize it.
- A mechanical ventilator should be used to ensure proper breathing for the patient. Provide.
- Provide the patient with the proper amount of fluid.
- Give the patient antibiotic medicine to protect him from infection.
- If the patient’s blood pressure increases, provide him with drugs to regulate it.
- If the patient has seizures, provide him with proper medicine.
- If the patient has nausea and If vomiting occurs, provide antiemetic medicine.
- Continuously monitor vital signs such as pulse, respiration, blood pressure, and body temperature.
- Provide treatment as needed if the patient has any other needs.
Nursing care:
- Check the patient’s vital signs.
- Check the patient’s pulse.
- Assess the patient’s ECG pattern.
- Monitor the patient’s arterial blood gas analysis.
- Monitor the patient’s serum electrolytes, as well as serum creatinine and blood urea nitrogen.
- Check the patient’s urine output hourly.
- Maintain the patient’s intake output chart.
- Check the patient’s heart sound, respiratory status and mental status.
- Check the patient’s vital signs.
- Do laboratory investigations on the patient.
- Check the patient’s level of consciousness. To do.
- Maintain the patient’s intake output chart.
- Prepare the patient if he has seizures or any other condition.
- Provide the patient with a comfortable and functional environment.
Define metabolic alkalosis:
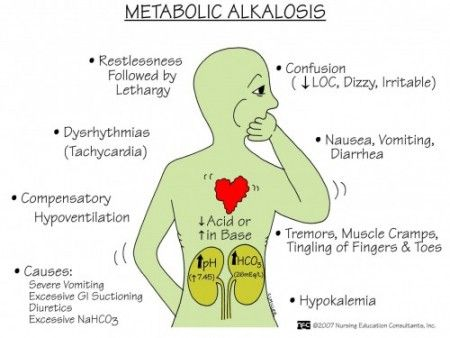
- Metabolic alkalosis is a condition in which the concentration of bicarbonate (Hco3-) and the concentration of pH (ph) are high. This is due to the gain of bicarbonate (Hco3-) and the loss of hydrogen (H+) ions.
- One of the main causes of metabolic alkalosis is vomiting and gastric suctioning, in which hydrogen and hydrogen ions are lost.
Etiology/ cause (Cause):
- Due to excessive accumulation of Hco3(Bicarbonate).
- Vomiting,
- Alkali administration,
- Nasogastric tube drainage or lavage.
- Pyloric Stenosis.
- Due to the use of steroid or diuretic medicines (furosemide, thiazide, and ethacrynic acid).
- External drainage of gastric fluids.
- Prolonged ingestion of milk and calcium carbonate.
- Hypercorticism.
- Hypercorticism.
- Hypercorticism Aldosteronism (hyperaldosteronism).
- (Cushing’s disease)
- Excessive ingestion of baking soda.
- Use of antacid medication.
- Administration of excessive amounts of intravenous fluids with a high bicarbonate concentration.
- Blood transfusion.
Clinical manifestation:
- Headache.
- Dizziness.
- Fatigue.
- Tingling sensation in fingers and toes.
- hypertonic muscles.
- neuromuscular excitability.
- delirium.
- tetany.
- stunning.
- difficulty breathing Padavi.
- tachycardia.
- Arrhythmias.
- Difficulty in breathing.
- cyanosis ( bluish discoloration of skin and nails).
- nausea,
- vomiting,
- diarrhea,
- irritability,
- confusion,
- increased heart rate,
- irregular heart beat,
- low blood pressure.
diagnostic evaluation (Diagnostic Evaluations):
- history taking and physical examination.
- Arterial blood gas evaluation, which is done by the P. pH level greater than 7.35.
- Serum bicarbonate concentration greater than 26 milliequivalents/liter (meq/liter).
- pco2(partial pressure of carbon dioxide) level over 45 mmhg.
- Assessment of urinary chloride level.
- serum electrolyte study shows low potassium,
Calcium And chloride level, - ECG finding sisclose alow t wave,merging with p wave,
Artirial and sinus tachycardia. - ct scan,
- MRI,
- renal angiography
Medical Management :
- Provide the patient with ammonium chloride (1 to 2 grams orally every 4 to 6 hours).
- Provide the patient with adequate chloride.
- Provide the patient with potassium chloride diluted in normal saline solution.
- To normalize the patient’s fluid volume, the patient should be given sodium chloride Administering.
- Keep the patient’s electrolyte balance abnormal.
- If the patient is being provided with diuretic therapy, stop it.
- If the patient has hypokalemia, administer potassium or KCl.
- If the patient has severe alkalosis, administer hydrochloric acid to the patient.
- Use carbonic anhydration to treat metabolic alkalosis.
- Administer hydrochloric acid intravenously 0.1 to 0.2 mmol/kg/liter to the patient.
- Maintain proper intake and output of the patient.
- If the kidneys are not functioning properly, then ask the patient to undergo dialysis to treat metabolic alkalosis.
- If the patient’s blood pressure increases If so, provide antihypertensive medicine.
- If the patient has nausea and vomiting, provide antiemetic medicine.
- Properly monitor the patient’s vital signs.
- Properly provide fluid and electrolytes to the patient.
Nursing Management :
- Monitor the patient’s respiratory rate, rhythm, and depth.
- Check the patient’s level of consciousness.
- Assess the patient’s skin color.
- Maintain the patient’s electrolyte balance.
- Prevent the patient from complications (siezer,coma)
- Provide a safe and noise-free environment to the patient.
- Provide side rails to the patient.
- Administer prescribed IV fluid to the patient.
- Check the patient’s arterial blood gas analysis and serum electrolytes.
- Check the patient’s vital signs and check peripheral pulse.
- Check the patient for any condition such as phlebitis at the IV site.
- Provide proper oxygen to the patient.
- Check the patient for muscle weakness, tetany, and decreased activity level.
- Maintain the patient’s intake output chart.
