P.B.B.Sc.F.Y-MICROBIOLOGY-2017-PAPER-no.1 (UPLOAD)(DONE)
P.B.B.Sc.F.Y-MICROBIOLOGY-PAPER SOLUTION-SAURASHTRA UNIVERSITY-GUJARAT-2017
SECTION-I
1 Long Essay: (any two) 2×10 = 20
(1) Define and Classify antibodies. Explain different antigen antibody Reaction. 10 MARKS
ANSWER :-
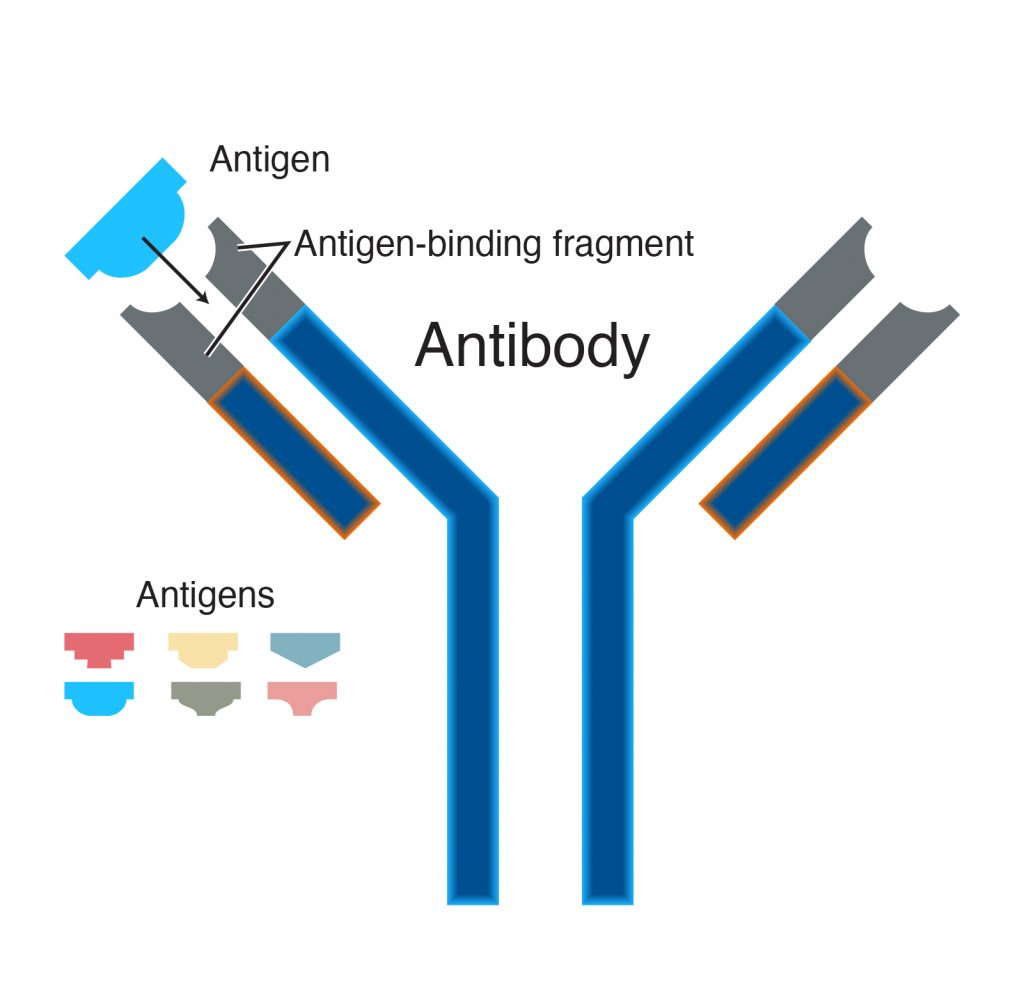
Define Antibodies
Antibodies, also known as immunoglobulins, are Y-shaped proteins produced by the immune system in response to the presence of foreign substances (antigens) such as viruses, bacteria, or toxins. Antibodies play a crucial role in the immune response by recognizing and binding to specific antigens, marking them for destruction by other immune cells or neutralizing their harmful effects directly. This process helps the body defend against pathogens and is a key aspect of adaptive immunity.
Classification of antibodies:
Immunoglobulin G (IgG):
- Structure: IgG is the most abundant antibody class in the bloodstream, making up about 75% of total antibodies in the body. It consists of two identical heavy chains and two identical light chains, forming a Y-shaped molecule.
- Function: IgG provides long-term immunity against bacterial and viral infections. It can cross the placenta, providing passive immunity to the fetus.
- Subclasses: IgG has four subclasses: IgG1, IgG2, IgG3, and IgG4, each with distinct functions and properties.
Immunoglobulin M (IgM):
- Structure: IgM is the largest antibody, composed of five units linked together by a J chain, forming a pentameric structure. Each unit has two heavy chains and two light chains.
- Function: IgM is the first antibody produced during an initial immune response. It is effective at agglutinating pathogens and activating the complement system.
Immunoglobulin A (IgA):
- Structure: IgA exists in two forms: monomeric IgA (found in the bloodstream) and dimeric IgA (predominantly found in mucosal secretions like saliva, tears, and breast milk). It consists of two heavy chains and two light chains.
- Function: IgA plays a crucial role in mucosal immunity by preventing pathogens from adhering to mucosal surfaces and neutralizing toxins and viruses.
Immunoglobulin D (IgD):
- Structure: IgD is found on the surface of B cells as a membrane-bound antibody. It consists of two heavy chains and two light chains.
- Function: IgD is involved in the activation of B cells, helping to initiate the immune response by recognizing antigens.
- Immunoglobulin E (IgE):
- Structure: IgE consists of two heavy chains and two light chains.
- Function: IgE is involved in allergic reactions and defense against parasites. It binds to mast cells and basophils, triggering the release of histamine and other inflammatory mediators.
types of antigen-antibody reactions:
Agglutination:
- Definition: Agglutination occurs when antibodies bind to multiple antigens, causing them to clump together.
- Mechanism: Antibodies have multiple binding sites, allowing them to cross-link antigens. This cross-linking leads to the formation of visible clumps or aggregates.
- Purpose: Agglutination helps immobilize pathogens, making it easier for immune cells to recognize and engulf them. It also enhances the efficiency of phagocytosis.
Precipitation:
- Definition: Precipitation occurs when antibodies bind to soluble antigens, forming immune complexes that become insoluble.
- Mechanism: Antibodies bind to antigens, forming antigen-antibody complexes. When these complexes reach a critical size, they become insoluble and precipitate out of solution.
- Purpose: Precipitation helps remove soluble antigens from bodily fluids by forming aggregates that can be engulfed and cleared by phagocytic cells.
Neutralization:
- Definition: Neutralization occurs when antibodies bind to toxins, viruses, or other harmful substances, preventing them from interacting with host cells.
- Mechanism: Antibodies bind to antigens, blocking their active sites or receptors. This prevents the antigens from exerting their harmful effects on host cells.
- Purpose: Neutralization helps protect the body from the harmful effects of pathogens and toxins by rendering them inactive and preventing further damage.
Opsonization:
- Definition: Opsonization is the process of tagging pathogens for destruction by immune cells such as macrophages and neutrophils.
- Mechanism: Antibodies bind to pathogens, coating them and making them more recognizable to phagocytic cells. This facilitates the recognition and engulfment of pathogens by immune cells.
- Purpose: Opsonization enhances the efficiency of phagocytosis, helping to clear pathogens from the body more effectively.
Complement Activation:
- Definition: Complement activation is a cascade of enzymatic reactions triggered by the binding of antibodies to antigens.
- Mechanism: Antibodies bound to antigens activate the complement system, leading to the recruitment of complement proteins and the formation of membrane attack complexes. These complexes can lyse pathogens, promote inflammation, and enhance phagocytosis.
- Purpose: Complement activation helps eliminate pathogens by directly lysing them, attracting immune cells to the site of infection, and enhancing the clearance of pathogens by phagocytic cells.
(2) Describe morphology, cultural characteristics and laboratory diagnosis of clostridium tetani. 10 marks
ANSWER :-
Clostridium tetani is a bacterium that causes tetanus, a potentially fatal disease characterized by muscle stiffness and spasms.
Here’s a detailed description of its morphology, cultural characteristics, and laboratory diagnosis:
Morphology:
- C. tetani is a Gram-positive, anaerobic, spore-forming bacillus.
- Under the microscope, it appears as long, slender rods with rounded ends.
- The bacterium forms round, terminal spores that give it a drumstick or tennis racket appearance.
Cultural Characteristics:
- C. tetani grows well under anaerobic conditions, meaning it thrives in environments devoid of oxygen.
- On solid media like blood agar, it forms small, round, opaque colonies with irregular edges.
- The colonies may exhibit a ground-glass appearance due to the accumulation of metabolic by-products.
- In liquid media, it produces a characteristic turbidity and gas production.
Laboratory Diagnosis:
- Clinical Symptoms: The diagnosis of tetanus often begins with clinical symptoms such as muscle stiffness, spasms, and difficulty swallowing or breathing.
- Microscopy: Gram staining of clinical specimens may reveal Gram-positive bacilli consistent with C. tetani, but this method alone is not sufficient for diagnosis.
- Culture: Culturing of samples on selective media under anaerobic conditions can isolate C. tetani. However, this process may take several days, and the yield can be low.
- Toxin Detection: C. tetani produces a potent neurotoxin called tetanospasmin, which is responsible for the symptoms of tetanus. Laboratory tests, such as enzyme-linked immunosorbent assay (ELISA), can detect tetanus toxin in serum or wound samples.
- Molecular Techniques: Polymerase chain reaction (PCR) assays targeting specific genes of C. tetani can provide rapid and sensitive identification of the bacterium in clinical samples.
- Serological Tests: Serological assays, including neutralization tests, can detect antibodies against tetanus toxin in serum samples, aiding in the diagnosis.
(3) Define Sterilization and explain in detail about methods of Sterilization.10 Marks
Define Sterilization
Sterilization is the process of killing or removing all forms of microorganisms, including bacteria, viruses, fungi, and spores, from an object or surface to prevent the transmission of infectious agents. There are various methods of sterilization, each with its advantages, limitations, and applications. Here are some common methods:
Methods of sterilization :-
- Heat Sterilization:
- Autoclaving: Autoclaving involves subjecting objects to high-pressure steam at temperatures typically ranging from 121°C to 134°C (250°F to 273°F) for a specified period, usually 15 to 30 minutes. Autoclaving is effective against a wide range of microorganisms, including bacteria, viruses, and spores, and is widely used in healthcare settings and laboratories.
- Dry Heat Sterilization: Dry heat sterilization utilizes hot air or flames to sterilize objects. It is less effective than autoclaving for killing spores but can be suitable for items that cannot withstand moist heat. Temperatures of 160°C to 180°C (320°F to 356°F) are typically used for several hours to achieve sterilization.
- Chemical Sterilization:
- Ethylene Oxide (ETO): Ethylene oxide is a gas used for the sterilization of heat-sensitive materials, such as plastics, electronics, and medical devices. It penetrates materials effectively and kills microorganisms by damaging their DNA and proteins. ETO requires specialized equipment and proper ventilation due to its toxic nature.
- Hydrogen Peroxide Gas Plasma: This method involves exposing objects to hydrogen peroxide vapor in a low-pressure plasma chamber. The plasma generates free radicals that destroy microbial structures. It is suitable for heat-sensitive items and is used in healthcare facilities.
- Radiation Sterilization:
- Ionizing Radiation: Ionizing radiation, such as gamma rays or electron beams, is used to sterilize medical devices, pharmaceuticals, and food products. It damages microbial DNA, preventing replication and causing cell death. Gamma radiation penetrates deeply and is commonly used for bulk sterilization, while electron beams are used for surface sterilization.
- Ultraviolet (UV) Radiation: UV radiation is used for surface sterilization of air, water, and surfaces in healthcare settings and laboratories. It damages microbial DNA, preventing replication. UV lamps are commonly used in biosafety cabinets, water treatment systems, and air disinfection units.
- Filtration:
- Membrane Filtration: Membrane filters with pore sizes smaller than the size of microorganisms are used to remove bacteria, fungi, and parasites from liquids, such as pharmaceuticals, vaccines, and biological fluids. Filtration is commonly used for heat-sensitive liquids or solutions that cannot be autoclaved.
- Other Methods:
- Chemical Disinfection: Chemical disinfectants, such as chlorine compounds, alcohols, and hydrogen peroxide, are used to disinfect surfaces, instruments, and medical devices. While these methods may not achieve sterilization, they can significantly reduce microbial contamination.
- Plasma Sterilization: Plasma sterilization uses low-temperature plasma to kill microorganisms on surfaces. It is effective for sterilizing heat-sensitive materials but requires specialized equipment.
Each sterilization method has its advantages and limitations, and the choice of method depends on factors such as the type of material to be sterilized, the level of microbial contamination, and the desired level of sterility. It is essential to follow proper protocols and validation procedures to ensure the effectiveness of the sterilization process.
2 . Short Essay (Any 3 out of 5)
(1) HIV
Human Immunodeficiency Virus (HIV) is a retrovirus that primarily targets cells of the human immune system, particularly CD4+ T cells and macrophages. HIV infection can lead to acquired immunodeficiency syndrome (AIDS), a condition characterized by a weakened immune system and increased susceptibility to opportunistic infections and certain cancers. Here are some key details about HIV:
- Structure: HIV is composed of an outer lipid envelope derived from the host cell membrane, which surrounds an inner core containing viral proteins and genetic material. The viral genome consists of two identical strands of single-stranded RNA. HIV also encodes several viral proteins, including structural proteins (e.g., Gag, Pol, and Env) and regulatory proteins (e.g., Tat and Rev).
- Transmission: HIV is primarily transmitted through contact with infected bodily fluids, including blood, semen, vaginal fluids, and breast milk. Common routes of transmission include unprotected sexual intercourse, sharing contaminated needles or syringes, and perinatal transmission from an HIV-positive mother to her child during pregnancy, childbirth, or breastfeeding.
- Lifecycle: The HIV replication cycle involves several stages, including viral entry, reverse transcription, integration, transcription, translation, assembly, and budding. During viral entry, HIV binds to CD4 receptors and co-receptors (e.g., CCR5 or CXCR4) on target cells, facilitating viral fusion and entry into the cell. Once inside the host cell, the viral RNA genome is reverse transcribed into double-stranded DNA by the enzyme reverse transcriptase. The viral DNA is then integrated into the host cell genome by the enzyme integrase, allowing for the transcription and translation of viral proteins. New viral particles are assembled and released from the host cell through budding, where they can infect other cells and continue the cycle of replication.
- Clinical Stages:
- Acute HIV Infection: The initial stage of HIV infection is characterized by a rapid increase in viral replication and a high concentration of virus in the blood. Acute HIV infection may cause flu-like symptoms, such as fever, fatigue, sore throat, and swollen lymph nodes, but can also be asymptomatic in some cases.
- Chronic HIV Infection: Without treatment, HIV infection progresses to chronic HIV infection, during which the immune system gradually deteriorates, leading to a decline in CD4+ T cell count and an increase in viral load.
- AIDS: AIDS is the most advanced stage of HIV infection, characterized by severe immune suppression and the development of opportunistic infections, malignancies, and other complications. The diagnosis of AIDS is based on the presence of specific opportunistic infections or a CD4+ T cell count below a certain threshold (<200 cells/mm³) in the absence of treatment.
- Diagnosis: HIV infection is diagnosed through serological testing, which detects antibodies to HIV in the blood, or molecular testing, which detects viral RNA or DNA. Common diagnostic tests include enzyme immunoassays (EIAs), rapid antibody tests, nucleic acid amplification tests (NAATs), and Western blot assays.
- Treatment: Antiretroviral therapy (ART) is the cornerstone of HIV treatment. ART consists of combinations of antiretroviral drugs that suppress viral replication, reduce viral load, and preserve immune function. Standard ART regimens typically include drugs from at least two different classes, such as nucleoside reverse transcriptase inhibitors (NRTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), protease inhibitors (PIs), integrase inhibitors (INSTIs), and entry inhibitors.
- Prevention: Prevention strategies for HIV include behavioral interventions (e.g., condom use, risk reduction counseling), biomedical interventions (e.g., pre-exposure prophylaxis [PrEP], post-exposure prophylaxis [PEP]), harm reduction programs (e.g., needle exchange programs), and treatment as prevention (TasP) initiatives. Promoting HIV testing, early diagnosis, and linkage to care are also essential components of HIV prevention efforts.
(2) Mycobacterium tuberculosis
Mycobacterium tuberculosis also known as Koch’s bacillus, is a species of pathogenic bacteria in the family Mycobacteriaceae and the causative agent of tuberculosis.First discovered in 1882 by Robert Koch, M.
👉Mycobacterium tuberculosis is the bacterium responsible for causing tuberculosis (TB), a potentially serious infectious disease that primarily affects the lungs but can also affect other parts of the body.
- Microbiology:
Mycobacterium tuberculosis is a rod-shaped, acid-fast bacterium. Acid-fastness refers to its ability to retain a dye when stained with acid-fast staining techniques, indicating a high lipid content in its cell wall.
- Transmission:
TB is primarily transmitted through the air when an infected individual coughs, sneezes, or speaks, releasing respiratory droplets containing the bacteria.
Close and prolonged contact with an infected person increases the risk of transmission.
- Pathogenesis: When inhaled, M. tuberculosis enters the lungs and is engulfed by macrophages, a type of immune cell. However, it can survive and replicate within these cells, evading the immune system. This leads to the formation of granulomas, which are collections of immune cells that wall off the bacteria. In some cases, the bacteria can remain dormant within these granulomas for years, leading to latent TB infection.
- Clinical Manifestations: TB can present as either active disease or latent infection.
In active TB, symptoms may include persistent cough, chest pain, fatigue, fever, night sweats, and weight loss.
In latent TB infection, individuals are asymptomatic and do not spread the disease, but they have the potential to develop active TB if their immune system becomes compromised.
- Diagnosis:
Diagnosis of TB often involves a combination of clinical evaluation, imaging studies (such as chest X-rays), microbiological tests (such as sputum smear microscopy, culture, and molecular tests like PCR), and tuberculin skin testing or interferon-gamma release assays to detect latent infection.
- Treatment:
TB is treated with a combination of antibiotics over a prolonged period (typically six to nine months) to ensure complete eradication of the bacteria.
Common antibiotics used include isoniazid, rifampicin, pyrazinamide, and ethambutol.
Drug-resistant TB strains, including multi-drug-resistant TB (MDR-TB) and extensively drug-resistant TB (XDR-TB), require specialized treatment regimens.
- Prevention: Prevention efforts focus on identifying and treating active cases promptly, as well as implementing measures to prevent transmission, such as infection control practices in healthcare settings, contact tracing, and vaccination with the Bacillus Calmette-Guérin (BCG) vaccine, although its efficacy varies.
(3) Bacterial growth curve
The bacterial growth curve typically consists of four phases: lag phase, log (exponential) phase, stationary phase, and death phase.
- Lag Phase:
Bacteria are adjusting to their environment, synthesizing enzymes, and preparing for growth.
There’s little to no increase in cell number during this phase.
- Log Phase:
Also known as the exponential phase, bacteria multiply rapidly, doubling in number at regular intervals.
This phase is characterized by balanced growth, with nutrients being plentiful and waste products not inhibiting growth.
- Stationary Phase:
Growth rate slows down due to depletion of nutrients, accumulation of waste products, and other factors.
The number of new cells produced equals the number of cells dying, resulting in a stable population size.
- Death Phase:
Conditions become unfavorable for bacterial growth, leading to a decline in the number of viable cells. The rate of cell death exceeds the rate of cell division.
(4) Hypersensitivity
Hypersensitivity refers to an exaggerated response by the immune system to a substance that is normally harmless, such as pollen, food proteins, or certain medications.
◾It’s categorized into four types:
- Type I hypersensitivity: Immediate allergic reactions involving IgE antibodies. Examples include allergic rhinitis (hay fever), asthma, and anaphylaxis.
- Type II hypersensitivity: Antibody-mediated reactions targeting specific cells or tissues. Examples include autoimmune hemolytic anemia and some drug reactions.
- Type III hypersensitivity: Immune complex-mediated reactions causing inflammation. Examples include serum sickness and certain autoimmune diseases like systemic lupus erythematosus.
- Type IV hypersensitivity: Delayed hypersensitivity reactions involving T cells. Examples include contact dermatitis and some drug reactions.
👉 Treatment and management
The treatment and management of hypersensitivity depend on the type and severity of the reaction. Here’s a general approach:
- Avoidance: Identify and avoid triggers whenever possible. This might involve avoiding certain foods, chemicals, or environmental factors.
- Medication: Depending on the type of hypersensitivity, medications such as antihistamines, corticosteroids, or epinephrine (for severe allergic reactions) may be prescribed.
- Allergen Immunotherapy: For allergies, allergen immunotherapy (such as allergy shots or sublingual immunotherapy) can help desensitize the body to specific allergens over time.
- Barrier Methods: For contact hypersensitivity, using barrier methods like gloves or clothing can prevent direct contact with irritants.
- Education: Educate yourself about your condition and triggers, and learn how to recognize and respond to reactions promptly.
- Lifestyle Changes: Making lifestyle changes such as improving indoor air quality, maintaining a healthy diet, and managing stress can help reduce the frequency and severity of reactions.
- Emergency Plan: If you have severe allergies, ensure you have an emergency action plan in place, including carrying epinephrine auto-injectors if prescribed.
(5) Culture media
ulture media in microbiology serve as a vital tool for cultivating and studying microorganisms in a controlled laboratory environment.
These media provide the necessary nutrients, pH levels, and other conditions required for the growth and isolation of microorganisms.
culture media in microbiology:
- Classification based on Physical State:
- Solid Media: These are agar-based and are used for isolating and culturing microorganisms as colonies on the surface of the medium.
- Liquid Media: These are broth-based and used primarily for the growth of microorganisms in suspension. They are often employed for large-scale growth or to assess microbial growth in liquid environments.
- Classification based on Composition:
- Chemically Defined Media: These media have precisely known compositions, with all components being chemically defined. They are advantageous for studying specific nutritional requirements of microorganisms.
- Complex Media: These media contain various organic and inorganic compounds of unknown composition, such as peptones, extracts, and meat infusions. They are more suitable for general-purpose growth of microorganisms and may support a broader range of organisms due to their complex nature.
- Classification based on Function:
- General Purpose Media: These media support the growth of a wide range of microorganisms and are often used for routine cultivation and maintenance of bacterial cultures. Examples include nutrient agar and tryptic soy agar.
- Selective Media: These media contain additives that inhibit the growth of certain organisms while allowing the growth of others. They are useful for isolating specific types of microorganisms from mixed cultures. Examples include MacConkey agar for selecting Gram-negative bacteria and Mannitol Salt agar for selecting staphylococci.
- Differential Media: These media allow for the differentiation of closely related organisms based on their biochemical characteristics. They typically contain indicators or substrates that produce visible changes in the presence of certain metabolic activities. Examples include blood agar, which distinguishes hemolytic patterns of bacteria based on their ability to lyse red blood cells.
- Enriched Media: These media contain additional nutrients, such as blood or serum, to support the growth of fastidious microorganisms with complex nutritional requirements. Examples include blood agar and chocolate agar, which are enriched with blood components.
- Specialized Media:
- Transport Media: These media are designed to maintain the viability of microorganisms during transport to the laboratory for analysis.
- Storage Media: These media are used for preserving cultures for extended periods, typically at low temperatures or in a frozen state.
- Preparation and Sterilization:
- Culture media are prepared by mixing the appropriate ingredients, adjusting the pH, and then sterilizing the medium by autoclaving or filtration to remove any contaminants.
- Agar is often added to solidify the medium, and after sterilization, it is poured into petri dishes or tubes for solid media or dispensed into containers for liquid media.
3 .Short Answers (Any 4 out of 6)-4×2 = 8
(1) Zeihl Neilsen’s staining
Answer :-

Ziehl-Neelsen staining
👉Ziehl-Neelsen staining, named after the German bacteriologist Franz Ziehl and the Norwegian physician Friedrich Neelsen, is a special staining technique primarily used to identify acid-fast bacteria, notably Mycobacterium tuberculosis, the causative agent of tuberculosis, as well as other species in the Mycobacterium genus.
Here’s a detailed breakdown of the staining procedure:
- Preparation of Smear: A sample suspected to contain acid-fast bacteria, such as sputum, tissue, or culture, is spread thinly on a microscope slide and allowed to air dry.
- Heat Fixation: The slide is gently heated to fix the bacterial cells onto the slide. This can be done by passing the slide through a flame or using a heating block.
- Primary Stain (Carbolfuchsin): The fixed smear is flooded with carbolfuchsin, a lipid-soluble dye containing basic fuchsin and phenol. The slide is then heated gently, often with steam, for several minutes. The heat helps to drive the stain into the mycolic acid-rich cell wall of acid-fast bacteria.
- Decolorization:
After staining, the slide is rinsed with acid-alcohol (a solution of hydrochloric acid in ethanol or methanol). Acid-fast bacteria retain the carbolfuchsin stain due to their waxy cell walls, while other bacteria are decolorized.
- Counterstain (Methylene Blue): To visualize non-acid-fast bacteria and background material, the slide is counterstained with methylene blue or a similar contrast stain. This step stains the decolorized cells blue.
- Mounting:
The slide is then rinsed with water to remove excess stain and air-dried. A coverslip is mounted over the smear using a mounting medium, such as Canada balsam or glycerol.
Under the microscope, acid-fast bacteria appear bright red or pink against a blue background, while non-acid-fast bacteria and other cellular material appear blue.
(2) Give four names of gram negative bacteria
Answer:- four examples of gram-negative bacteria:
- Escherichia coli (E. coli)
- Salmonella spp.
- Pseudomonas aeruginosa
- Neisseria gonorrhoeae
(3) Pasteurization
Answer:-
Pasteurization is a process of heating liquid food, usually milk or juice, to a specific temperature for a set period to kill pathogens without significantly altering the taste or nutritional value.
It’s named after Louis Pasteur, the French scientist who developed the process in the 19th century.
- Purpose: The primary goal of pasteurization is to destroy harmful bacteria, viruses, and other microorganisms present in food and beverages, particularly in liquids like milk, juice, beer, and wine.
- Process: The process involves heating the liquid to a specific temperature for a set period and then rapidly cooling it.
- Temperature and Time: milk is typically pasteurized at a minimum temperature of 161°F (71.7°C) for at least 15 seconds, while in some other countries, the standards may differ slightly.
- Methods: There are two main methods of pasteurization:
- High-Temperature Short-Time (HTST) pasteurization: This method involves heating the liquid to a high temperature (usually around 161°F or 71.7°C) for a short period (15-20 seconds) before rapidly cooling it.
- Effects on Food: Pasteurization kills most harmful microorganisms while preserving the taste, texture, and nutritional value of the liquid.
- Safety: While pasteurization significantly reduces the risk of foodborne illnesses, it does not guarantee complete sterilization.
(4) Chemotherapy
Answer:-
chemotherapy
Chemotherapy is a treatment that uses drugs to kill cancer cells or stop them from growing and dividing. These drugs can be administered orally or intravenously and work throughout the body, targeting cancer cells wherever they may be.
- Administration: Chemotherapy can be given in different ways, including intravenously (through a vein), orally (in pill or liquid form), or through injections into muscles or directly into the cancer site (intrathecal chemotherapy).
- Treatment Schedule: Chemotherapy is often given in cycles, with a period of treatment followed by a period of rest to allow the body to recover.
- Side Effects: While chemotherapy can be effective in killing cancer cells, it can also cause side effects because it affects normal cells as well. Common side effects include nausea, vomiting, hair loss, fatigue, and increased risk of infections. However, these side effects can often be managed with medication and other supportive measures.
- Combination Therapies: Chemotherapy is frequently used in combination with other treatments, such as surgery, radiation therapy, or targeted therapy, to improve outcomes for cancer patients. This approach, known as multimodal therapy, can increase the effectiveness of treatment and reduce the risk of cancer recurrence.
- Effectiveness: The effectiveness of chemotherapy varies depending on the type and stage of cancer, as well as individual factors such as overall health and response to treatment.
(5) ETO
Ethylene oxide (ETO) is a highly reactive gas that is used for sterilization in microbiology and various other industries. Its effectiveness in killing microorganisms makes it a valuable tool for ensuring the safety of medical devices, pharmaceuticals, and other sensitive materials.
👉ETO’s use in microbiology:
- Sterilization Process: ETO sterilization is typically carried out in a controlled environment, such as a sterilization chamber.
- Mechanism of Action: ETO is a powerful alkylating agent, meaning it disrupts the DNA of microorganisms, preventing their replication and causing their death.
- Compatibility: One of the main advantages of ETO sterilization is its compatibility with a wide range of materials, including plastics, rubber, metals, and electronic components.
- Residuals: One concern with ETO sterilization is the potential for residual gas and by-products remaining on sterilized items.
- Regulation and Safety: Due to its toxicity and potential carcinogenicity, ETO sterilization processes are highly regulated.
- Alternatives: While ETO is effective, there is ongoing research and development of alternative sterilization methods that are safer and more environmentally friendly, such as hydrogen peroxide gas plasma sterilization and supercritical carbon dioxide sterilization.
(6) CFT
The Complement Fixation Test (CFT) is a laboratory technique used to detect the presence of specific antibodies in a patient’s serum against a particular antigen, typically an infectious agent such as a bacterium or virus. It is based on the principle of the complement system’s ability to lyse cells when activated in the presence of antibody-antigen complexes.
Here’s how the Complement Fixation Test generally works:
- Preparation: The test begins by collecting a serum sample from the patient suspected of being infected with a particular pathogen. The serum is separated from the blood cells and can contain antibodies that the patient’s immune system has produced in response to the infection.
- Antigen and Complement: The specific antigen related to the suspected infection is mixed with the patient’s serum. If antibodies to the antigen are present in the serum, they will bind to the antigen, forming immune complexes.
- Addition of Complement: After allowing time for antibody-antigen complexes to form, complement proteins are added to the mixture. If complement-fixing antibodies are present in the patient’s serum, they will bind to the antigen, forming immune complexes that activate the complement system.
- Complement Activation: If the patient’s serum contains antibodies specific to the antigen, the complement system will be activated in a cascade of reactions. This activation leads to the lysis of sheep or guinea pig red blood cells that have been added to the mixture.
- Detection: The degree of hemolysis (breakdown of red blood cells) is observed. If complement-fixing antibodies are present in the patient’s serum, they will consume the complement proteins in the immune complexes, preventing hemolysis. Thus, the absence of hemolysis indicates a positive reaction, indicating the presence of specific antibodies against the antigen in the patient’s serum.
The Complement Fixation Test is a sensitive and specific assay for detecting antibodies in patient sera, but it is relatively complex and labor-intensive compared to other serological tests such as enzyme-linked immunosorbent assays (ELISAs) or immunofluorescence assays (IFAs). Despite this, it has been historically used for diagnosing various infectious diseases, including bacterial infections like syphilis and brucellosis, as well as viral infections such as influenza and measles.
SECTION – II
1.Long Essay (any 1 out of 2). 1×10 = 1
(1)
(a) Define HIV? 02
HIV stands for Human Immunodeficiency Virus.
◾ It’s a virus that attacks the immune system, specifically targeting CD4 cells, weakening the body’s ability to fight infections and diseases.
(b) Describe structure, clinical features and lab diagnosis of HIV.
Answer:-
1. Structure of HIV:
- HIV is a retrovirus with a complex structure. It contains an outer lipid envelope derived from the host cell membrane.
- Inside the envelope, there is a protein capsid that encloses the viral RNA genome and associated proteins.
- The viral genome consists of two identical strands of single-stranded RNA.
- HIV also encodes various viral proteins, including structural proteins (e.g., Gag, Pol, and Env) and regulatory proteins (e.g., Tat and Rev).
2. Clinical Features of HIV:
- The clinical presentation of HIV infection can vary depending on the stage of the disease.
- Acute HIV Infection: Often presents with flu-like symptoms, such as fever, fatigue, sore throat, swollen lymph nodes, and rash. However, some individuals may be asymptomatic.
- Chronic HIV Infection: As the infection progresses, symptoms may include persistent or recurrent infections, weight loss, diarrhea, night sweats, and fatigue.
- AIDS: The most advanced stage of HIV infection, characterized by severe immune suppression and the development of opportunistic infections (e.g., Pneumocystis pneumonia, tuberculosis, cryptococcal meningitis), malignancies (e.g., Kaposi’s sarcoma, non-Hodgkin lymphoma), and other complications. Without treatment, AIDS is often fatal.
3. Laboratory Diagnosis of HIV:
- Serological Testing: Serological tests detect antibodies to HIV in the blood. Common serological tests include enzyme immunoassays (EIAs), rapid antibody tests, and Western blot assays. These tests are typically used for screening and diagnosis.
- Molecular Testing: Molecular tests detect viral RNA or DNA in the blood. Nucleic acid amplification tests (NAATs), such as polymerase chain reaction (PCR) assays, are used to detect HIV RNA or DNA. These tests are highly sensitive and are often used for early diagnosis, monitoring viral load, and detecting HIV in infants born to HIV-positive mothers.
- CD4+ T Cell Count: Monitoring CD4+ T cell count is an essential component of HIV management. A decrease in CD4+ T cell count indicates immune system suppression and disease progression.
- Viral Load Measurement: Viral load testing quantifies the amount of HIV RNA in the blood. It is used to monitor the effectiveness of antiretroviral therapy (ART) and assess disease progression.
Early diagnosis of HIV is crucial for initiating timely treatment and preventing the progression of the disease to AIDS. Testing and diagnosis should be followed by linkage to care, initiation of antiretroviral therapy, and ongoing monitoring of HIV viral load and CD4+ T cell count to optimize outcomes for individuals living with HIV.
(a) Define bio-medical waste.02
Biomedical waste refers to any kind of waste that contains potentially infectious materials, such as blood or bodily fluids.
It includes items like needles, syringes, bandages, and other materials used in medical procedures.
Proper disposal of biomedical waste is crucial to prevent the spread of infections and protect public health and the environment.
(b) Explain about classification and methods of disposal of different categories of biomedical waste.08
👉Biomedical waste is classified into different categories based on its source, type, and potential risk of infection.
The World Health Organization (WHO) and various national health agencies typically provide guidelines for this classification. The categories generally include:
- General Biomedical Waste (GBW): This category includes non-hazardous biomedical waste that doesn’t pose a significant risk of infection. It comprises items like paper, cardboard, and non-infectious materials used in healthcare settings.
- Hazardous Biomedical Waste (HBW): HBW includes waste materials that are potentially infectious or hazardous to human health or the environment. This category encompasses items like used needles, syringes, cultures, and stocks of infectious agents.
- Sharps Waste: This category specifically refers to items that can puncture or lacerate the skin, such as needles, syringes, lancets, and broken glass. Sharps waste poses a high risk of transmitting blood-borne pathogens.
- Infectious Waste: Infectious waste contains materials that are known or suspected to be infected with pathogens, including blood, bodily fluids, cultures, and tissues. Proper disposal is crucial to prevent the spread of infectious diseases.
- Pathological Waste: This category includes human tissues, organs, body parts, and fluids removed during surgery, autopsy, or medical procedures. Pathological waste requires careful handling and disposal to prevent contamination and potential health risks.
👉Methods of disposal for biomedical waste vary depending on the type and local regulations, but common methods include:
- Incineration: This is one of the most effective methods for disposing of biomedical waste, particularly for sharps, cultures, and pathological waste. Incineration involves burning the waste at high temperatures to destroy pathogens and reduce the volume of waste.
- Autoclaving: Autoclaving uses steam under pressure to sterilize biomedical waste, killing microorganisms and rendering the waste safe for disposal. After autoclaving, the waste can often be safely disposed of in landfills.
- Chemical Treatment: Some biomedical waste, such as liquid waste or certain laboratory materials, can be treated with chemical disinfectants to neutralize pathogens before disposal. Chemical treatment may be followed by other disposal methods like landfilling or sewage treatment.
- Microwave Treatment: Microwave technology can be used to disinfect biomedical waste by heating it to high temperatures. This method is effective for certain types of waste but may not be suitable for all materials.
- Encapsulation: This method involves encapsulating biomedical waste in solid materials such as cement or resin to contain any pathogens and prevent their release into the environment. Encapsulation is often used for sharps and other solid waste.
2 .Short Essay (any 3 out of 5) 3×5 = 15
(1) Principles of microbiology
Answer :-
Microbiology is the study of microorganisms, including bacteria, viruses, fungi, protozoa, and algae. It delves into their structure, function, reproduction, metabolism, and genetics. Key principles include:
- Cellular Structure: Microorganisms vary in cell structure, such as bacteria with cell walls and viruses with protein coats. Understanding these structures aids in identifying and classifying microorganisms.
- Metabolism: Microbes exhibit diverse metabolic pathways, including aerobic respiration, anaerobic respiration, and fermentation. These pathways impact their growth, survival, and interactions with their environment.
- Growth and Reproduction: Microorganisms can reproduce rapidly through processes like binary fission, budding, or spore formation. Factors like temperature, pH, and nutrient availability influence their growth rates.
- Genetics: Microbial genetics explores how genes are transferred, expressed, and regulated within microbial populations. This includes mechanisms like mutation, conjugation, transformation, and transduction.
- Ecology and Diversity: Microorganisms inhabit diverse environments, from soil and water to human bodies. Their roles in ecosystems, such as nutrient cycling and symbiotic relationships, are crucial for understanding microbial ecology.
- Pathogenesis and Immunity: Some microorganisms cause disease in hosts, leading to the study of pathogenesis and host immune responses. Understanding these interactions is essential for developing treatments and vaccines.
- Biotechnology: Microorganisms have extensive applications in biotechnology, including producing antibiotics, enzymes, vaccines, and genetically modified organisms (GMOs).
- Microbial Control: Principles of microbiology guide efforts to control and manage microbial populations, including sanitation, sterilization, and the use of antimicrobial agents.
- Microbial Evolution: Microorganisms evolve rapidly, leading to the emergence of new strains and resistance to antimicrobial drugs. Studying microbial evolution informs strategies for combating resistance and understanding evolutionary processes.
(2) Bacterial nutrition
Answer:-
Bacteria exhibit a diverse range of nutritional strategies, allowing them to thrive in various environments, from soil and water to the human body. Their nutritional requirements can be categorized into several main types:
Autotrophic Nutrition:
- Photoautotrophy: Some bacteria, such as cyanobacteria, are capable of harnessing light energy to produce organic compounds from inorganic substances (e.g., carbon dioxide). They contain pigments like chlorophyll that enable them to perform photosynthesis.
- Chemoautotrophy: Certain bacteria can utilize inorganic compounds, such as hydrogen sulfide or ammonia, as sources of energy to fix carbon dioxide and synthesize organic molecules. Examples include sulfur-oxidizing bacteria and nitrifying bacteria.
- Heterotrophic Nutrition:
- Saprophytic: Saprophytic bacteria decompose dead organic matter, such as dead plants or animals, and derive nutrients from the breakdown products. They play a crucial role in nutrient cycling and decomposition processes in ecosystems.
- Parasitic: Parasitic bacteria obtain nutrients from living host organisms, often causing harm or disease in the process. They may reside intracellularly or extracellularly within the host and can cause a wide range of infectious diseases in humans and other animals.
- Symbiotic: Symbiotic bacteria establish mutually beneficial relationships with host organisms. For example, some bacteria live in the guts of animals and assist in digestion (e.g., gut microbiota), while others form symbiotic associations with plant roots and aid in nitrogen fixation (e.g., rhizobia).
Mixotrophic Nutrition:
- Some bacteria are capable of utilizing both organic and inorganic sources of carbon and energy, exhibiting a mixotrophic nutritional strategy. This versatility allows them to adapt to changing environmental conditions and optimize their nutrient acquisition.
In addition to their energy and carbon sources, bacteria require various essential nutrients, including nitrogen, phosphorus, sulfur, and trace elements, for growth and metabolism. They obtain these nutrients from their surrounding environment, often competing with other organisms for limited resources.
(3) Morphology, and laboratory diagnosis of haemophilus influenza.
Morphology of Haemophilus influenzae:*
Answer :-
- H. influenzae is a small, pleomorphic, gram-negative coccobacillus.
- It appears as tiny, non-motile rods or cocci, often occurring singly or in pairs.
- It is facultatively anaerobic, meaning it can grow with or without oxygen.
- H. influenzae requires factors X (hemin) and V (nicotinamide adenine dinucleotide, NAD) for growth, which can be provided by blood agar or chocolate agar.
Laboratory Diagnosis:
Culture:
- H. influenzae is cultured on chocolate agar or blood agar plates in an atmosphere enriched with 5-10% carbon dioxide.
- Colonies typically appear as small, smooth, greyish-white colonies after 24-48 hours of incubation.
- Satellitism: H. influenzae colonies often exhibit satellitism when grown adjacent to Staphylococcus aureus colonies, due to the release of factor V by S. aureus.
Gram Staining:
- H. influenzae stains gram-negative, appearing as pink/red cocci or rods under the microscope.
Biochemical Tests:
- H. influenzae is catalase-positive and oxidase-positive.
- It does not ferment carbohydrates.
Serological Tests:
- Serotyping of H. influenzae can be done based on the presence of polysaccharide capsule antigens.
- The most clinically significant serotypes are a through f.
Molecular Methods:
- Polymerase Chain Reaction (PCR) can be used for the rapid and specific detection of H. influenzae DNA.
Antibiotic Susceptibility Testing:
- H. influenzae should be tested for antibiotic susceptibility, as resistance to various antibiotics can occur.
Clinical Samples:
- Clinical samples for diagnosis include blood, cerebrospinal fluid (CSF), sputum, throat swabs, and other appropriate specimens depending on the suspected site of infection.
Diagnostic Imaging:
- In cases of suspected invasive disease, such as meningitis or epiglottitis, imaging studies like CT scans or MRI may be performed to assess the extent of infection.
(4) Immunity
Answer :-
Immunity refers to the ability of an organism to resist and defend against harmful pathogens, such as bacteria, viruses, parasites, and other foreign substances. It is a vital component of the body’s defense mechanisms and plays a crucial role in maintaining overall health and well-being. There are two main types of immunity: innate immunity and adaptive immunity.
- Innate Immunity:
- Natural or Nonspecific Immunity: Innate immunity is the first line of defense against pathogens and provides immediate, nonspecific protection. It includes physical and chemical barriers, such as the skin and mucous membranes, as well as cellular components like phagocytes (e.g., macrophages, neutrophils) and natural killer (NK) cells. Innate immunity recognizes common patterns associated with pathogens, known as pathogen-associated molecular patterns (PAMPs), through pattern recognition receptors (PRRs). This type of immunity is present from birth and does not provide long-lasting protection against specific pathogens.
- Adaptive Immunity:
- Acquired or Specific Immunity: Adaptive immunity is a highly specialized defense mechanism that provides targeted and long-lasting protection against specific pathogens. It is characterized by the ability to recognize and remember specific antigens associated with pathogens. Adaptive immunity involves two main components:
- Cell-mediated Immunity: Mediated by T lymphocytes (T cells), which recognize antigens presented by infected or abnormal cells. Cell-mediated immunity is critical for combating intracellular pathogens, such as viruses and certain bacteria, as well as for coordinating immune responses.
- Humoral Immunity: Mediated by B lymphocytes (B cells) and antibodies (immunoglobulins). B cells produce antibodies that bind to specific antigens, marking them for destruction by other immune cells or neutralizing their harmful effects. Humoral immunity is essential for combating extracellular pathogens, such as bacteria and toxins.
Adaptive immunity exhibits immunological memory, meaning that upon initial exposure to a specific pathogen, the immune system generates a robust response and “remembers” the pathogen’s antigens. Upon subsequent encounters with the same pathogen, the immune response is faster and more effective due to the presence of memory T and B cells.
(5) Tetanus
Answer:-
Tetanus, also known as lockjaw, is a serious bacterial infection caused by the bacterium Clostridium tetani. The bacterium produces a toxin called tetanospasmin, which affects the nervous system, leading to muscle stiffness and spasms. Here are some key details about tetanus:
1. Transmission: Tetanus bacteria are typically found in soil, dust, and animal feces. The bacteria can enter the body through open wounds or cuts, particularly deep puncture wounds, burns, or surgical wounds. Tetanus is not transmitted from person to person.
2. Pathogenesis:
- After entering the body, the tetanus bacteria release tetanospasmin toxin.
- The toxin travels via nerves to the central nervous system (CNS), where it blocks the release of inhibitory neurotransmitters, such as glycine and gamma-aminobutyric acid (GABA).
- Without the inhibitory signals, there is uncontrolled stimulation of motor neurons, leading to muscle spasms and rigidity characteristic of tetanus.
3. Clinical Features:
- The hallmark symptom of tetanus is muscle stiffness and spasms, particularly in the jaw muscles (hence the term “lockjaw”).
- Other symptoms may include stiffness and spasms of neck muscles (resulting in difficulty swallowing), facial muscles, back muscles (resulting in arching of the back), abdominal muscles (resulting in abdominal rigidity), and limb muscles.
- In severe cases, respiratory muscles can be affected, leading to difficulty breathing and potentially life-threatening respiratory failure.
- Symptoms typically develop within days to weeks after exposure to the bacteria.
4. Diagnosis:
- Tetanus is primarily diagnosed based on clinical symptoms and history of exposure to the bacteria (e.g., wound or injury).
- Laboratory tests, such as culture of wound samples or detection of tetanus toxin in serum, are not typically performed for diagnosis but may be used in research settings.
5. Prevention:
- Tetanus can be prevented through vaccination with the tetanus toxoid vaccine, often administered as part of the combination vaccine known as DTaP (diphtheria, tetanus, acellular pertussis) for children or Tdap (tetanus, diphtheria, acellular pertussis) for adolescents and adults.
- Routine booster doses of tetanus toxoid vaccine are recommended every 10 years for adults to maintain immunity.
6. Treatment:
- Treatment of tetanus involves wound care to remove the source of infection and administration of tetanus immune globulin (TIG) to neutralize circulating toxin.
- Supportive care, including muscle relaxants, sedatives, and mechanical ventilation, may be necessary to manage muscle spasms and respiratory complications.
- Antibiotics, such as metronidazole or penicillin, are given to eliminate the bacteria from the body.
Tetanus is a potentially life-threatening condition, but it is preventable through vaccination and proper wound care. Prompt medical attention is crucial for the management of tetanus, particularly in severe cases.
3 . Short Answers (Compulsory) 6×2 = 12
(1) Complement system
Answer:-
The complement system is a part of the immune system that consists of a group of proteins circulating in the blood and tissues. It plays a crucial role in defending the body against pathogens, such as bacteria, viruses, and other microorganisms, as well as in the clearance of immune complexes and dead cells.
- Recognition and Activation: The complement system can be activated through three different pathways:
- Classical Pathway: Activated by antibodies bound to antigens on the surface of pathogens. The binding of C1 complex to these antibody-antigen complexes initiates a cascade of enzymatic reactions.
- Alternative Pathway: Constitutively active at a low level and can be spontaneously activated by certain molecular patterns on pathogens.
- Lectin Pathway: Triggered by the binding of pattern recognition molecules, such as mannose-binding lectin (MBL), to specific carbohydrate patterns on pathogens.
- Cascade Amplification: Once activated, the complement system undergoes a cascade of enzymatic reactions, resulting in the cleavage and activation of complement proteins. This cascade amplifies the initial signal, leading to a robust immune response.
- Effector Functions:
- Opsonization: Complement proteins coat the surface of pathogens, enhancing their recognition and phagocytosis by immune cells such as macrophages and neutrophils.
- Inflammation: Complement activation leads to the release of inflammatory mediators, such as histamine and cytokines, promoting vasodilation, increased vascular permeability, and recruitment of immune cells to the site of infection.
- Membrane Attack Complex (MAC) Formation: Terminal complement proteins assemble to form the MAC, which creates pores in the membranes of target cells, causing them to lyse and die.
- Regulation: To prevent excessive or inappropriate activation, the complement system is tightly regulated by various inhibitors and regulators. These molecules ensure that complement activation is targeted specifically to pathogens and does not cause damage to host tissues.
(2) Ameobiasis
Answer :-
👉Amoebiasis, also known as amebiasis, is an infectious disease caused by the amoeba parasite, specifically Entamoeba histolytica.
This parasite is commonly found in contaminated food and water
Symptoms of amoebiasis can include:
- Diarrhea, which may be bloody
- Stomach cramps or abdominal pain
- Fatigue
- Weight loss
- Fever
- Nausea and vomiting
Diagnosis of amoebiasis usually involves stool sample analysis to detect the presence of the parasite or its DNA.
Treatment for amoebiasis typically involves antibiotics to kill the parasite. Commonly prescribed medications include metronidazole or tinidazole, which are effective in eliminating the parasite from the body.
Prevention of amoebiasis primarily involves:
- Drinking clean, safe water
- Washing hands frequently.
- Properly washing and cooking food, particularly fruits and vegetables
- Avoiding consumption of untreated water or raw foods in areas with poor sanitation.
(3) Montoux test
Answer:-
he Montoux test, also known as the Mantoux test, is a diagnostic tool used to determine whether someone has been exposed to the bacterium that causes tuberculosis (TB).
It involves injecting a small amount of purified protein derivative (PPD), a substance derived from the TB bacterium, into the skin, usually on the forearm.
After 48 to 72 hours, a healthcare professional will examine the injection site for a raised, red bump.
The size of the bump is measured to determine the result.
A positive result indicates that the person has been exposed to TB bacteria, either through infection or vaccination in the past, but it does not necessarily mean they have active TB disease.
Further evaluation, such as chest X-rays and sputum tests, may be needed to confirm TB infection or disease.
(4) Cell mediated immune response
Answer :-
The cell-mediated immune response involves T cells identifying and destroying infected or abnormal cells in the body. T cells recognize specific antigens presented on the surface of infected cells and then activate to eliminate the threat.
This response is crucial for combating intracellular pathogens like viruses and certain bacteria, as well as for identifying and destroying cancerous cells.
Involved in the cell-mediated immune response:
- Antigen Presentation:
- T Cell Activation:
- Cytokine Release:
- Differentiation of Effector T Cells:
- Cytotoxic T Cell Action:
- Memory T Cell Formation: .
(5) ELISA test
Elisa, or enzyme-linked immunosorbent assay, is a common laboratory technique
It used to detect the presence of antibodies or antigens in a sample.
It involves multiple steps, including coating a plate with the target molecule, adding the sample, washing to remove unbound material, adding a detection antibody, and then measuring the signal.
The results are typically quantified by color change or fluorescence. Elisa tests are widely used in medical diagnostics, research, and quality control.
(6) Types of immunoglobulin.
Answer :-
IgG (Immunoglobulin G):
- IgG is the most abundant type of antibody in the bloodstream, making up about 75% of all antibodies in the body.
- It provides long-term immunity against viruses, bacteria, and toxins.
- IgG can cross the placenta, providing passive immunity to the fetus.
IgM (Immunoglobulin M):
- IgM is the first antibody produced in response to an infection.
- It’s found mainly in the bloodstream and lymph fluid.
- IgM is effective at agglutinating (clumping together) pathogens, making them easier for other immune cells to destroy.
IgA (Immunoglobulin A):
- IgA is found predominantly in bodily fluids such as saliva, tears, breast milk, and mucus.
- It provides protection against infections in mucosal areas, such as the respiratory and gastrointestinal tracts.
- IgA helps prevent pathogens from attaching to mucous membranes.
IgE (Immunoglobulin E):
- IgE is involved in allergic reactions and defense against parasites.
- It triggers the release of histamine and other chemicals that cause allergic symptoms, such as sneezing, itching, and swelling.
- IgE levels are often elevated in individuals with allergies or parasitic infections.
IgD (Immunoglobulin D):
- IgD is found in small amounts in the bloodstream.
- Its exact function isn’t fully understood, but it’s believed to play a role in activating B cells.
- IgD may also be involved in immune regulation and defense against pathogens