First Year Post Basic B. Sc. (Nursing)-February 2019-Microbiology(done)(UPLOAD PAPER NO.5)
Microbiology–February 2019
SECTION – 1
1 Long Essays: (Answer Any Two) 2×10-20
💛 (a) Which is the causative agent of tetanus? Describe the morphology, modes of transmission and lab. Diagnosis of this agent. How tetanus can be prevented ?
The causative agent of tetanus is Clostridium tetani. This bacterium is an anaerobic, spore-forming, Gram-positive rod. The disease is primarily caused by the neurotoxin tetanospasmin, which is produced by Clostridium tetani. This potent toxin interferes with the normal functioning of the nervous system, leading to the characteristic muscle stiffness and spasms associated with tetanus. The spores of Clostridium tetani are commonly found in soil, dust, and animal feces, and infection typically occurs through contamination of wounds with these spores.
- Morphology*: Clostridium tetani is a gram-positive, anaerobic, spore-forming bacillus. It has a distinctive drumstick appearance under the microscope due to its terminal spores.
- Modes of Transmission: Tetanus spores are commonly found in soil, dust, and animal feces. The bacterium enters the body through wounds or cuts, especially puncture wounds, burns, and compound fractures. It can also enter through surgical procedures, intravenous drug use, or animal bites.
- Laboratory Diagnosis: Laboratory diagnosis involves isolating and identifying the bacterium from wound specimens. This is typically done using culture techniques under anaerobic conditions. The presence of the bacterium’s characteristic spores can also be observed in clinical samples under the microscope. Molecular techniques such as PCR (polymerase chain reaction) may also be used for detection.
- Prevention:
- Vaccination: Tetanus can be effectively prevented through vaccination with the tetanus toxoid-containing vaccine (TT vaccine). This vaccine is usually given as part of the childhood vaccination schedule and is often administered in combination with other vaccines like diphtheria and pertussis (DTaP vaccine).
- Booster Doses: Booster doses of the tetanus vaccine are recommended every 10 years for adults to maintain immunity. In cases of wounds or injuries, a tetanus booster may be administered if it has been more than 5 years since the last dose.
- Wound Care: Proper wound care is crucial to prevent tetanus. Thoroughly cleaning and disinfecting wounds can help remove tetanus spores and reduce the risk of infection.
- Passive Immunization: In cases of suspected tetanus exposure in individuals who are not adequately immunized, passive immunization with tetanus immune globulin (TIG) may be administered to provide immediate protection.
💛 (b) Enumerate the causative agents of urinary tract infection (UTI). What are the predisposing factors for UTI? Describe various specimen collection methods. How the specimen is processed in the laboratory?
Causative Agents of Urinary Tract Infections (UTIs):
- Escherichia coli (E. coli): This bacterium is the most common cause of UTIs, responsible for about 80% of cases.
- Staphylococcus saprophyticus: Another common bacterium, particularly in young sexually active women.
- Klebsiella pneumoniae, Proteus mirabilis, Enterococcus faecalis: These are other bacteria that can cause UTIs, albeit less frequently.
- Fungal Infections: Yeasts such as Candida albicans can also cause UTIs, especially in individuals with compromised immune systems.
Predisposing Factors for UTIs:
- Gender: Women are more prone to UTIs due to their shorter urethra, making it easier for bacteria to reach the bladder.
- Sexual Activity: Sexual intercourse can introduce bacteria into the urinary tract.
- Urinary Catheterization: Catheter use increases the risk of UTIs as it can introduce bacteria into the urinary tract.
- Anatomy: Certain anatomical factors such as urinary tract abnormalities can predispose individuals to UTIs.
- Menopause: Changes in estrogen levels can affect the vaginal flora, increasing susceptibility to UTIs in postmenopausal women.
- Immune System: Immunocompromised individuals are at higher risk of UTIs.
- Urinary Tract Obstruction: Any obstruction in the urinary tract can lead to stasis of urine, providing an environment conducive to bacterial growth.
Various Specimens in the Laboratory:
In the laboratory, various specimens are collected for the diagnosis of UTIs, including:
- Midstream Clean-Catch Urine: This is the most common method of urine collection to avoid contamination from the genital area.
- Catheterized Urine: In cases where a patient is unable to provide a clean-catch sample, urine can be obtained directly from the bladder via a catheter.
- Suprapubic Aspiration: This involves inserting a needle directly into the bladder through the abdominal wall to obtain a urine sample, usually done under ultrasound guidance.
- Urine Culture: A urine sample is streaked onto culture plates and incubated to identify the causative agent and determine its susceptibility to antibiotics.
- Urinalysis: This involves analyzing the physical, chemical, and microscopic properties of urine, including the presence of white blood cells, red blood cells, bacteria, and other substances indicative of infection or inflammation.
💛 (c) Classify Biomedical wastes. Write methods of segregation, treatment and disposal of Biomedical wastes.
Biomedical wastes can be classified into several categories:
- Infectious Waste: Waste that contains pathogens, such as blood or bodily fluids.
- Sharps: Needles, scalpels, or other sharp objects that may cause injuries or infections.
- Pathological Waste: Human tissues, organs, or body parts.
- Pharmaceutical Waste: Expired or unused medications, vaccines, or chemotherapy drugs.
- Chemical Waste: Chemicals used in healthcare facilities, like disinfectants or solvents.
- Radioactive Waste: Materials contaminated with radioactive substances, like nuclear medicine materials.
Methods of Segregation:
- Color-Coded Bins: Separate bins for different types of waste, each color-coded for easy identification.
- Labeling: Clearly label containers to indicate the type of waste they contain.
- Training: Educate staff on proper segregation techniques to ensure compliance.
Treatment of Biological Waste:
- Autoclaving: Steam sterilization method that kills pathogens by exposure to high temperature and pressure.
- Incineration: Controlled burning of waste at high temperatures to reduce volume and destroy pathogens.
- Chemical Treatment: Using disinfectants or chemical agents to neutralize pathogens.
- Microwave Treatment: Heating waste in a microwave to kill bacteria and viruses.
Disposal of Biological Waste:
- Landfill Disposal: Non-infectious biomedical waste can be disposed of in sanitary landfills.
- Hazardous Waste Facilities: Infectious or hazardous biomedical waste may require specialized facilities for disposal.
- Waste-to-Energy Plants: Some facilities can incinerate biomedical waste to generate energy.
- On-Site Treatment: Some healthcare facilities have their own treatment systems to process biomedical waste on-site.
2 Short Essays (Answer Any Three) 3×5=15
💛 (a) Hot air oven
A hot air oven is a type of dry heat sterilization equipment used in laboratories and medical facilities to sterilize materials and equipment. Here’s a detailed overview:
Description and Function
A hot air oven uses dry heat to sterilize materials. It operates by circulating hot air at high temperatures for a specified period, effectively killing microorganisms, including bacteria, viruses, and spores, by oxidation.
Key Features
- Temperature Range: Typically operates at temperatures between 50°C and 300°C, with common sterilization temperatures being 160°C, 170°C, or 180°C.
- Timer: Allows users to set specific sterilization periods, usually ranging from 1 to 2 hours depending on the temperature and nature of the material.
- Air Circulation: Equipped with fans or blowers to ensure even distribution of hot air, which is crucial for effective sterilization.
- Insulation: Designed with proper insulation to maintain consistent internal temperatures and energy efficiency.
Uses
- Sterilization of Laboratory Glassware: Including petri dishes, pipettes, flasks, and test tubes.
- Sterilization of Metal Instruments: Such as scalpels, forceps, and scissors.
- Heat-Resistant Materials: Sterilizes items that can withstand high temperatures without degradation, including some plastics and powders.
- Non-Aqueous Materials: Effective for sterilizing non-water-based materials that may not be suitable for autoclave sterilization.
Advantages
- Effective Sterilization: Ensures complete destruction of microorganisms and spores.
- Non-Corrosive: Unlike steam sterilization (autoclaving), dry heat does not corrode metal instruments.
- No Moisture: Suitable for materials that may be damaged by moisture.
Disadvantages
- Longer Cycle Times: Compared to autoclaves, hot air ovens typically require longer sterilization times.
- High Temperatures: Not suitable for heat-sensitive materials, which may be damaged by the high temperatures.
- Energy Consumption: Can be energy-intensive due to the high temperatures required.
Sterilization Protocols
- Preparation: Ensure items are clean and dry. Arrange them in the oven so that hot air can circulate freely around each item.
- Temperature and Time: Common protocols include:
- 160°C for 2 hours
- 170°C for 1 hour
- 180°C for 30 minutes
- Cooling: Allow items to cool gradually in the oven to prevent thermal shock or damage.
Maintenance and Safety
- Regular Calibration: Ensure accurate temperature control and uniform heat distribution.
- Cleaning: Regular cleaning to prevent contamination and buildup of residues.
- Safety Precautions: Use heat-resistant gloves and handle sterilized items with care to avoid burns.
💛 (b) Agglutination reactions
- Definition: Agglutination is the clumping together of particles, typically cells or bacteria, in the presence of specific antibodies or other substances.
- Process:
- When antibodies encounter their specific antigens (proteins or other molecules on the surface of cells or bacteria), they bind to them.
- As more antibodies bind to multiple antigens on different particles, they form bridges between the particles, causing them to clump together.
- Antibodies: Agglutination reactions are commonly used in blood typing and immunology tests. For example, blood typing determines the blood group based on the presence or absence of specific antigens on red blood cells, which agglutinate in the presence of corresponding antibodies.
- Applications:
- Blood typing: ABO blood group system and Rh factor.
- Bacterial identification: Agglutination assays are used to identify and classify bacteria based on their surface antigens.
- Serological tests: Such as latex agglutination tests for detecting antibodies or antigens in patient samples.
- Types of Agglutination:
- Direct Agglutination: Agglutination occurs when antibodies directly bind to antigens on the surface of particles.
- Indirect Agglutination: A secondary molecule, such as latex beads or red blood cells coated with antigen, is used to detect the presence of antibodies in a sample.
- Diagnostic Significance: The agglutination reaction is widely used in diagnostic tests due to its simplicity, specificity, and sensitivity. It allows for rapid detection and identification of specific antigens or antibodies in various biological samples.
💛 (c) Lab. diagnosis of filariasis
laboratory diagnosis of filariasis:
- Clinical Examination: Initial diagnosis often begins with clinical examination, where symptoms such as lymphedema, hydrocele, or swelling of limbs are observed.
- Microscopic Examination of Blood Smears: This is the most common method. A drop of blood is collected typically between 10 pm to 2 am (since microfilariae are more likely to be circulating in peripheral blood during this time). The blood smear is stained with Giemsa stain and examined under a microscope for the presence of microfilariae.
- Concentration Techniques: Concentration techniques like Knott’s concentration method or membrane filtration are used to enhance detection sensitivity by concentrating microfilariae from large volumes of blood.
- Serological Tests: Immunological methods like enzyme-linked immunosorbent assay (ELISA) or immunochromatographic tests (ICT) detect specific antibodies or antigens associated with filarial infection. These tests are useful when microfilariae are not present in blood.
- Molecular Techniques: Polymerase chain reaction (PCR) assays can detect filarial DNA in blood samples with high sensitivity and specificity. PCR is particularly useful for detecting low levels of infection and for species differentiation.
- Ultrasonography: Ultrasonography can be used to detect adult worms in the lymphatic system or detect associated conditions like hydrocele or lymphedema.
- X-ray: X-ray imaging may be used to visualize calcified adult worms in the lymphatic vessels.
- Skin Snip Test: This method involves taking a small skin snip from the affected area and examining it under a microscope for the presence of microfilariae or other parasite stages.
- Antigen Detection in Urine: Some antigen detection tests can detect filarial antigens in urine samples, providing an alternative non-invasive method for diagnosis.
- Histopathology: In cases where adult worms are surgically removed, histopathological examination of the tissues can confirm the diagnosis.
💛 (d) Kyasanur forest disease
Kyasanur Forest Disease (KFD):
- Overview: Kyasanur Forest Disease (KFD), also known as monkey fever, is a viral hemorrhagic fever caused by the Kyasanur Forest Disease Virus (KFDV), a member of the Flavivirus genus in the Flaviviridae family.
- Geographical Distribution: KFD is endemic to certain regions of India, primarily in the Western Ghats of Karnataka, Maharashtra, Kerala, and Tamil Nadu. It’s named after the Kyasanur Forest area in Karnataka where it was first identified in 1957.
- *Vector: The primary vector of KFDV is the tick species *Haemaphysalis spinigera. Ticks become infected by feeding on infected animals, primarily small mammals like rodents and shrews, as well as monkeys. Humans are incidental hosts and can become infected through tick bites.
- Clinical Presentation: KFD typically presents with symptoms similar to other viral hemorrhagic fevers, including fever, headache, myalgia (muscle pain), vomiting, diarrhea, and hemorrhagic manifestations such as petechiae, ecchymosis, and bleeding from mucous membranes. Severe cases can lead to neurological symptoms, including encephalitis.
- Incubation Period: The incubation period for KFD is typically 3 to 8 days after the tick bite.
- Diagnosis:
- Laboratory diagnosis involves detecting viral RNA in blood or serum samples using techniques like reverse transcription-polymerase chain reaction (RT-PCR).
- Serological tests such as enzyme-linked immunosorbent assay (ELISA) can detect antibodies against KFDV.
- Prevention:
- Vaccination: A formalin-inactivated tissue culture vaccine is available for individuals at high risk of exposure, such as forest workers and healthcare workers in endemic areas.
- Avoiding tick bites by wearing protective clothing, using insect repellents, and avoiding known tick-infested areas.
- Vector control measures, including environmental management to reduce tick habitats and use of acaricides (tick-killing chemicals).
- Treatment:
- Supportive care: There is no specific antiviral treatment for KFD. Treatment is primarily supportive, including hydration, pain management, and management of complications.
- Ribavirin, an antiviral drug, has shown some efficacy in early stages of the disease.
- Epidemiology: KFD outbreaks typically occur in rural forested areas during the dry season when tick activity is highest. Human cases often cluster around forested regions where infected ticks are prevalent.
- Public Health Importance: KFD can have significant public health implications, particularly in rural communities where outbreaks can lead to morbidity and mortality among both humans and primates. Surveillance, early detection, and prompt response are essential for controlling outbreaks.
💛 (e) Shigella
Shigella:
- Overview: Shigella is a genus of bacteria that causes a severe form of diarrhea known as shigellosis. There are four species of Shigella: S. dysenteriae, S. flexneri, S. boydii, and S. sonnei.
- Transmission: Shigella is primarily spread through the fecal-oral route, usually via contaminated food or water, or through direct contact with an infected person.
- Symptoms: Shigellosis typically presents with diarrhea, often bloody, along with abdominal pain, fever, and sometimes vomiting. The severity of symptoms can vary.
- Pathogenesis: Shigella bacteria invade the cells lining the colon and cause inflammation, leading to the characteristic symptoms of shigellosis. They produce toxins that contribute to the severity of the disease.
- Diagnosis: Diagnosis is usually made by culturing a stool sample to identify the presence of Shigella bacteria. Polymerase chain reaction (PCR) tests can also be used for diagnosis.
- Treatment: Antibiotics are often prescribed to treat shigellosis, although resistance to commonly used antibiotics has become a concern in some regions. Supportive care, such as fluid replacement therapy, is also important, especially in cases of severe dehydration.
- Prevention: Preventing Shigella infection involves practicing good hygiene, including thorough handwashing with soap and water, especially after using the bathroom or changing diapers. Proper food handling and sanitation are also crucial.
- Epidemiology: Shigellosis is more common in developing countries with poor sanitation infrastructure, but outbreaks can occur anywhere. It can affect people of all ages, but children are particularly susceptible.
- Complications: In severe cases, shigellosis can lead to complications such as dehydration, electrolyte imbalances, and in rare cases, reactive arthritis or hemolytic uremic syndrome (HUS).
- Control Measures: Public health measures to control Shigella infections include surveillance, outbreak investigations, education on hygiene practices, and efforts to improve sanitation and access to clean water.
3 Short Answers (Answer Any One) 1×3=3
💛 (a) Mycetoma
- Definition: Mycetoma, also known as Madura foot, is a chronic granulomatous subcutaneous infection caused by various fungi (eumycetoma) or bacteria (actinomycetoma).
- Epidemiology: Mycetoma is endemic in tropical and subtropical regions, particularly in Africa, Asia, and South America, where barefoot walking is common.
- Etiology: It can be caused by various organisms, including fungi such as Madurella, Scedosporium, and bacteria such as Actinomadura, Nocardia.
- Transmission: Entry of the causative organism occurs through skin trauma, typically through puncture wounds, leading to the development of localized infection.
- Clinical Presentation: Mycetoma typically presents as painless swelling and sinuses in subcutaneous tissues, commonly affecting the feet, although it can occur in other parts of the body. Sinuses may produce grains (aggregates of organisms) that vary in color depending on the causative organism.
- Diagnosis: Diagnosis involves a combination of clinical presentation, imaging studies (X-ray, MRI, ultrasound), and microbiological examination of grains obtained from sinuses or tissue biopsy.
- Treatment: Treatment usually involves long-term antimicrobial therapy based on the identified organism’s sensitivity, combined with surgical debridement to remove infected tissue and sinuses.
- Prognosis: Prognosis depends on the extent of the infection, the causative organism, and the timeliness of treatment. Early diagnosis and appropriate management can prevent complications such as tissue destruction and disability.
- Prevention: Prevention strategies include wearing protective footwear, particularly in endemic areas, and prompt treatment of any skin injuries to prevent infection.
💛 (b) Rubella
- Overview: Rubella, also known as German measles, is a contagious viral infection caused by the rubella virus. It’s usually mild in children, but it can be dangerous for pregnant women, potentially causing serious birth defects or miscarriage if contracted during pregnancy.
- Transmission: Rubella spreads through respiratory droplets when an infected person coughs or sneezes. It can also spread through direct contact with an infected person’s respiratory secretions.
- Symptoms: Symptoms typically appear 2 to 3 weeks after exposure and may include a mild fever, sore throat, swollen lymph nodes, headache, runny nose, and a fine, pink rash that starts on the face and spreads to the rest of the body.
- Complications: While rubella is usually mild, it can lead to complications, especially in pregnant women. The most serious complication is congenital rubella syndrome (CRS), which can cause severe birth defects such as deafness, blindness, heart defects, and intellectual disabilities.
- Diagnosis: Rubella is diagnosed based on symptoms and confirmed with a blood test that detects antibodies to the virus.
- Treatment: There is no specific treatment for rubella, as it’s a viral infection. Treatment typically focuses on relieving symptoms, such as rest, fluids, and over-the-counter pain relievers.
- Prevention: The best way to prevent rubella is through vaccination. The measles, mumps, and rubella (MMR) vaccine is highly effective in preventing rubella and its complications. It’s usually given as two doses, starting in childhood.
- Vaccination during pregnancy: Rubella vaccination is not recommended during pregnancy because the vaccine contains live, weakened virus and could potentially harm the fetus. Women should be vaccinated before becoming pregnant to ensure protection.
SECTION -11
4 Long Essays: (Answer Any One)1×10-10
💛 (a) Which is the causative organism of diphtheria? Describe its morphology, pathogenesis and lab, diagnosis.
- Causative Organism*: Corynebacterium diphtheriae, a gram-positive, rod-shaped bacterium.
- Morphology:
- C. diphtheriae appears as irregularly stained, club-shaped rods with tapered ends.
- It often forms palisades or Chinese letter arrangements in culture.
- Some strains may exhibit metachromatic granules when stained with methylene blue.
- Pathogenesis:
- The primary virulence factor is diphtheria toxin, encoded by a bacteriophage within the bacterial genome.
- Upon infection, C. diphtheriae colonizes the respiratory tract, typically the pharynx and tonsils.
- Diphtheria toxin inhibits protein synthesis in host cells, leading to tissue damage and formation of a characteristic pseudomembrane composed of dead tissue, fibrin, and inflammatory cells.
- Toxin may also disseminate systemically, causing myocarditis, neuritis, and other complications.
- Laboratory Diagnosis:
- Microscopy: Gram staining reveals gram-positive rods with characteristic morphology.
- Culture: Throat swabs are plated onto selective media like Löffler’s medium or tellurite agar, which suppresses the growth of other bacteria. C. diphtheriae typically forms grayish-black colonies within 24-48 hours.
- Biochemical Tests: Catalase-positive, urease-negative, and non-motile. May exhibit metachromatic granules.
- Toxin Detection: Polymerase chain reaction (PCR) assays can detect the presence of the toxin gene (tox) or toxin production.
- Serology: Antitoxin antibodies can be detected in serum samples of infected individuals.
- Elek Test: A confirmatory test where C. diphtheriae colonies are streaked on a special medium, and the presence of toxin is indicated by a precipitation line.
- Treatment:
- Administer diphtheria antitoxin to neutralize circulating toxin.
- Antibiotics such as erythromycin or penicillin are given to eradicate the bacteria.
- Supportive care to manage airway, cardiac, and neurological complications.
💛 (b) Enumerate four routes by which hepatitis B virus can spread. Draw a labelled schematic diagram of the virus. Describe briefly on the viral markers that appear during hepatitis B infection and prophylaxis available for the virus.
Schematic Diagram of Hepatitis B Virus:
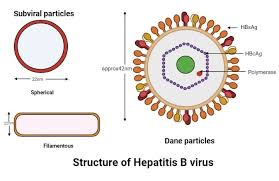
Routes of Hepatitis B Virus (HBV) Transmission:
- Parenteral Transmission:
- Through percutaneous exposure to infected blood or body fluids, such as through needlestick injuries, sharing needles or syringes, or receiving contaminated blood transfusions or organ transplants.
- Vertical Transmission:
- From mother to child during childbirth, when the infant comes into contact with maternal blood or vaginal secretions containing HBV.
- Sexual Transmission:
- Through unprotected sexual contact with an infected partner, particularly through vaginal, anal, or oral sex.
- Perinatal Transmission:
- From an infected mother to her infant during pregnancy or through breast milk.
Viral markers that appear during hepatitis B infection
- Viral Markers during Hepatitis B Infection:
- HBsAg (Hepatitis B surface antigen): Appears early in infection and is a marker of active HBV infection.
- HBcAg (Hepatitis B core antigen): Not detectable in serum but can be detected in liver tissue during acute infection.
- HBeAg (Hepatitis B e antigen): Indicates active viral replication and high infectivity.
- Anti-HBs (Hepatitis B surface antibody): Appears during recovery or as a result of vaccination, indicating immunity to HBV.
- Anti-HBc (Hepatitis B core antibody): Appears during or after acute infection, indicating past or ongoing infection.
Prophylaxis and Treatment:
- Vaccination:
- Hepatitis B vaccine is highly effective in preventing HBV infection and is recommended for all infants, healthcare workers, individuals at high risk of exposure, and those with certain medical conditions.
- Post-Exposure Prophylaxis:
- Individuals exposed to HBV, such as through needlestick injuries or sexual contact with an infected person, may receive post-exposure prophylaxis with hepatitis B immune globulin (HBIG) and the hepatitis B vaccine to prevent infection.
- Antiviral Treatment:
- Antiviral medications, such as nucleoside/nucleotide analogs (e.g., tenofovir, entecavir), are available to suppress HBV replication and reduce the risk of liver damage in individuals with chronic HBV infection.
- Screening and Counseling:
- Routine screening for HBV infection, especially in high-risk populations, allows for early detection and treatment, as well as counseling on prevention measures to reduce transmission.
5 Short Essays (Answer Any Three) 3×5-15
💛 (a) Autoclave
An autoclave is a device used to sterilize equipment and materials using high-pressure saturated steam. It is a vital tool in medical, laboratory, and industrial settings to ensure that instruments and other items are free of microorganisms.
Description and Function
An autoclave uses steam at high pressure and temperature to achieve sterilization. The standard conditions for autoclaving are 121°C (250°F) at 15 psi for a specified time, usually 15-20 minutes, depending on the nature and volume of the material being sterilized.
Key Components
- Chamber: The space where items to be sterilized are placed. It is usually made of stainless steel.
- Jacket: Surrounds the chamber and is filled with steam to help maintain the chamber temperature.
- Steam Generator: Produces the steam needed for the sterilization process.
- Control System: Regulates temperature, pressure, and time to ensure proper sterilization.
- Safety Valve: Prevents overpressure by releasing excess steam.
- Pressure Gauge: Monitors the pressure inside the chamber.
- Thermometer: Monitors the temperature inside the chamber.
Sterilization Cycle
- Loading: Items are loaded into the autoclave in a way that allows steam to circulate freely around them.
- Pre-Vacuum (optional): Air is removed from the chamber to ensure steam penetration.
- Heating: Steam is introduced into the chamber, and temperature and pressure begin to rise.
- Sterilization: The chamber reaches the desired temperature and pressure, maintained for the required time to achieve sterilization.
- Exhaust: Steam is vented from the chamber, and pressure is reduced.
- Drying (optional): A drying cycle may be included to remove moisture from sterilized items.
- Cooling: Items are allowed to cool before they are removed from the autoclave.
Types of Autoclaves
- Gravity Displacement Autoclave: Uses gravity to displace air with steam. Suitable for solid and uncomplicated loads.
- Pre-Vacuum Autoclave: Removes air from the chamber before introducing steam, ensuring better penetration. Suitable for porous and complex loads.
- Benchtop Autoclave: Smaller, used for sterilizing small loads, often in laboratories and dental clinics.
- Vertical Autoclave: Compact design, often used in laboratories with limited space.
- Horizontal Autoclave: Larger capacity, used in hospitals and research facilities.
Uses
- Medical and Surgical Instruments: Sterilizes instruments used in surgeries and medical procedures.
- Laboratory Glassware and Media: Sterilizes glassware, growth media, and other lab materials.
- Biohazardous Waste: Sterilizes waste materials to render them safe for disposal.
- Industrial Applications: Used in manufacturing processes where sterile conditions are necessary.
Maintenance and Safety
- Regular Maintenance: Routine inspection and maintenance of seals, valves, and the chamber to ensure proper function.
- Calibration: Periodic calibration of temperature and pressure sensors.
- Cleaning: Regular cleaning of the chamber to remove residue and prevent contamination.
- Safety Training: Ensuring all users are trained in the safe operation of the autoclave, including loading procedures and emergency shutdown.
Advantages
- Effective Sterilization: Highly effective in killing all forms of microbial life, including spores.
- Versatility: Can sterilize a wide variety of materials and instruments.
- Speed: Relatively quick process compared to other sterilization methods.
Disadvantages
- Heat Sensitivity: Not suitable for materials that cannot withstand high temperatures and pressure.
- Energy Consumption: High energy use due to the need for heating and maintaining pressure.
Autoclaves are essential devices in healthcare, laboratories, and various industries for ensuring the sterility of instruments and materials. By using high-pressure steam, autoclaves effectively eliminate all forms of microbial life, contributing to the prevention of infections and contamination. Proper use, maintenance, and safety procedures are crucial for maximizing the efficiency and lifespan of an autoclave.
💛 (b) Prevention of hospital acquired infections
Prevention of hospital-acquired infections (HAIs) is a critical aspect of patient safety and healthcare quality. Here are key strategies and best practices to prevent HAIs:
1. Hand Hygiene
- Frequent Hand Washing: Use soap and water or an alcohol-based hand sanitizer before and after patient contact, after touching potentially contaminated surfaces, and after removing gloves.
- Education and Training: Regular training sessions for healthcare workers on proper hand hygiene techniques and the importance of compliance.
2. Use of Personal Protective Equipment (PPE)
- Appropriate Use of PPE: Gloves, gowns, masks, and eye protection should be used as required by the type of patient interaction or procedure.
- Proper Donning and Doffing: Training on correct methods to put on and remove PPE to avoid self-contamination.
3. Sterilization and Disinfection
- Equipment Sterilization: Proper sterilization of surgical instruments, reusable equipment, and devices.
- Surface Disinfection: Regular cleaning and disinfection of patient rooms, operating theaters, and common areas with hospital-grade disinfectants.
4. Safe Injection Practices
- Use of Sterile Equipment: Always use sterile needles and syringes. Never reuse needles or syringes.
- Single-Use Vials: Prefer single-dose vials to multiple-dose vials when possible.
5. Environmental Hygiene
- Regular Cleaning Protocols: Establish and follow protocols for cleaning and disinfecting surfaces, especially high-touch areas like doorknobs, bed rails, and light switches.
- Monitoring and Auditing: Regular audits to ensure compliance with cleaning protocols.
6. Antimicrobial Stewardship
- Prudent Use of Antibiotics: Implement policies for the appropriate use of antibiotics to reduce the development of resistant strains of bacteria.
- Review and Feedback: Regular review of antibiotic prescriptions and feedback to prescribers.
7. Infection Control Policies and Procedures
- Isolation Precautions: Implement contact, droplet, or airborne precautions for patients with known or suspected infections.
- Standard Precautions: Use standard precautions for all patients, assuming that every person is potentially infected or colonized with an organism that could be transmitted.
8. Surveillance and Reporting
- Infection Surveillance: Regular monitoring and reporting of infection rates and outbreaks within the hospital.
- Feedback Mechanisms: Provide feedback to healthcare workers on infection rates and trends to encourage adherence to preventive measures.
9. Education and Training
- Ongoing Education: Continuous education and training programs for healthcare workers on infection prevention and control practices.
- Patient and Family Education: Educate patients and their families about infection prevention measures and encourage them to speak up if they notice lapses in infection control practices.
10. Implementation of Bundles
- Care Bundles: Use evidence-based care bundles for common procedures associated with HAIs, such as central line-associated bloodstream infections (CLABSIs), catheter-associated urinary tract infections (CAUTIs), and ventilator-associated pneumonia (VAP).
11. Engineering Controls
- Ventilation Systems: Proper maintenance of heating, ventilation, and air conditioning (HVAC) systems to ensure adequate air exchange and filtration.
- Hand Hygiene Facilities: Ensure easy access to hand washing stations and hand sanitizer dispensers throughout the facility.
12. Patient Care Practices
- Catheter Care: Use catheters only when necessary and ensure proper insertion and maintenance techniques.
- Surgical Site Infection (SSI) Prevention: Follow protocols for preoperative skin antisepsis, appropriate use of prophylactic antibiotics, and proper postoperative wound care.
💛 (c) Type I hypersensitivity reactions
Type I hypersensitivity reaction, also known as immediate hypersensitivity or allergic reaction, is an immune response triggered by the binding of allergens to IgE antibodies on mast cells and basophils, leading to the release of inflammatory mediators. Here are the key points:
- Initiation:
- Allergens, such as pollen, dust mites, certain foods, or insect venom, enter the body through inhalation, ingestion, injection, or contact with the skin.
- Antigen-presenting cells (APCs) present these allergens to T-helper cells, which stimulate B cells to produce IgE antibodies specific to the allergen.
- Sensitization:
- IgE antibodies produced in response to the allergen bind to high-affinity receptors (FcεRI) on the surface of mast cells and basophils, sensitizing them.
- Activation:
- Upon re-exposure to the allergen, it binds to IgE antibodies on sensitized mast cells and basophils, cross-linking the IgE receptors.
- This cross-linking triggers mast cells and basophils to release preformed inflammatory mediators such as histamine, leukotrienes, and prostaglandins.
- Inflammatory Response:
- Histamine and other mediators cause vasodilation, increased vascular permeability, smooth muscle contraction, and mucus secretion.
- These effects lead to the characteristic symptoms of allergic reactions, including itching, hives, rhinitis, bronchoconstriction, gastrointestinal disturbances, and in severe cases, anaphylaxis.
- Clinical Manifestations:
- Type I hypersensitivity reactions can manifest as allergic rhinitis (hay fever), allergic asthma, atopic dermatitis (eczema), food allergies, insect sting allergies, and drug allergies.
- Treatment:
- Management of type I hypersensitivity reactions involves allergen avoidance when possible.
- Pharmacological treatments include antihistamines, corticosteroids, bronchodilators, and epinephrine (in cases of anaphylaxis).
- Allergen immunotherapy (desensitization) may be recommended for individuals with severe allergies to induce tolerance to specific allergens.
- Prevention:
- Identifying and avoiding triggers is crucial in preventing allergic reactions.
- Education about allergen avoidance, proper use of medications (e.g., inhalers, epinephrine auto-injectors), and emergency action plans are important components of managing allergies and preventing severe reactions.
💛 (d) Lab. diagnosis of pulmonary tuberculosis
- Microscopic Examination:
- Acid-fast bacilli (AFB) staining: A sputum sample is stained with a dye such as Ziehl-Neelsen or auramine-rhodamine and examined under a microscope for the presence of acid-fast bacilli, which are characteristic of Mycobacterium tuberculosis.
- Culture:
- Sputum culture: Sputum samples are cultured on specialized media such as Lowenstein-Jensen or Middlebrook agar to isolate and grow Mycobacterium tuberculosis bacteria.
- Culturing allows for the definitive identification of the bacteria and also provides information about drug susceptibility, which is crucial for guiding treatment.
- Nucleic Acid Amplification Tests (NAATs):
- Polymerase chain reaction (PCR) assays: NAATs detect the genetic material (DNA or RNA) of Mycobacterium tuberculosis in sputum samples, providing rapid and sensitive diagnosis, especially in cases where microscopy and culture may be negative or delayed.
- Examples include the GeneXpert MTB/RIF assay, which simultaneously detects M. tuberculosis and rifampicin resistance within a few hours.
- Serological Tests:
- Serological tests detect antibodies produced by the immune system in response to M. tuberculosis infection.
- However, these tests are generally not recommended for diagnosing pulmonary tuberculosis due to their low sensitivity and specificity.
- Molecular Typing:
- Molecular typing techniques, such as DNA fingerprinting or genotyping, can be used for epidemiological studies and to track the transmission of specific strains of M. tuberculosis within populations.
- Radiological Imaging:
- While not a laboratory test, chest X-rays or CT scans are often used as adjuncts to laboratory diagnosis to detect characteristic pulmonary changes associated with tuberculosis, such as infiltrates, cavities, and lymphadenopathy.
- Histopathological Examination:
- In cases where sputum samples are not available or when extra-pulmonary tuberculosis is suspected, biopsy specimens from affected tissues (e.g., lymph nodes, lung tissue) may be examined under a microscope for the presence of granulomas and acid-fast bacilli.
- Clinical Correlation:
- Laboratory results should always be interpreted in conjunction with clinical findings, radiological imaging, and risk factors for tuberculosis to arrive at a comprehensive diagnosis and treatment plan.
- Follow-up Monitoring:
- Regular monitoring of sputum smears and cultures during treatment is essential to assess treatment response and detect drug resistance early.
💛 (e) Lab. diagnosis of enteric fever
- Blood Cultures*:
- Blood cultures are the gold standard for diagnosing enteric fever.
- Multiple blood samples are collected over a 24-hour period to increase the chances of isolating the causative organism.
- The collected samples are inoculated into specialized culture media, such as BACTEC or blood agar, and incubated for several days to allow the growth of bacteria.
- The presence of Salmonella typhi or Salmonella paratyphi in blood cultures confirms the diagnosis of enteric fever.
- Stool Cultures:
- Stool cultures may also be performed to isolate Salmonella typhi or Salmonella paratyphi from fecal samples.
- However, stool cultures have lower sensitivity compared to blood cultures and may be negative, especially in the later stages of the disease.
- Bone Marrow Culture:
- Bone marrow culture is more sensitive than blood culture and may be considered, especially when blood cultures are negative but clinical suspicion for enteric fever remains high.
- Bone marrow aspiration is performed, and the aspirate is inoculated into culture media for bacterial growth.
- Serological Tests:
- Serological tests, such as Widal test and Typhidot, detect antibodies produced in response to Salmonella typhi or Salmonella paratyphi infection.
- However, these tests have limitations in terms of sensitivity, specificity, and cross-reactivity with other infections, and their results should be interpreted cautiously.
- Molecular Tests:
- Polymerase chain reaction (PCR) assays can detect the genetic material of Salmonella typhi or Salmonella paratyphi directly from clinical specimens, such as blood or stool.
- PCR assays offer rapid and sensitive diagnosis, especially in cases where bacterial cultures may be negative or delayed.
- Urinary Antigen Tests:
- Some commercial tests are available to detect Salmonella typhi antigens in urine samples.
- These tests offer rapid results and may be useful as adjuncts to blood cultures, especially in resource-limited settings.
- Clinical Correlation:
- Laboratory results should always be interpreted in the context of clinical findings, including symptoms such as fever, abdominal pain, and gastrointestinal symptoms, as well as travel history to endemic regions.
- Antibiotic Sensitivity Testing:
- Antimicrobial susceptibility testing should be performed on isolated strains to guide appropriate antibiotic therapy, especially due to the emergence of antimicrobial resistance in Salmonella typhi and Salmonella paratyphi strains.
6 Short Answers (Answer All Questions) 2×6=12
💛 (a) Candidiasis
💛(b) Gas gangrene
💛 (c) Bacterial flagella
Bacterial Flagella:
- Structure: Flagella are long, whip-like appendages found on the surface of some bacteria, enabling motility.
- Composition: They are composed of a protein called flagellin arranged in a helical fashion around a hollow core.
- Function: Flagella rotate like propellers, allowing bacteria to move in response to chemical signals or environmental cues, aiding in navigation towards favorable conditions or away from harmful substances.
💛 (d) Cutaneous anthrax
Cutaneous Anthrax:
👉1. Bacterial Infection:
Cutaneous anthrax is the most common form of anthrax infection and is caused by the bacterium Bacillus anthracis.
- Transmission: It typically occurs through direct contact with spores from contaminated animal products, such as wool, hides, or meat, or through contact with contaminated soil.
- Clinical Presentation: Cutaneous anthrax presents as a localized skin lesion with a characteristic painless ulcer that progresses to a black eschar (a thick, black scab) surrounded by edema and redness. Systemic symptoms are usually absent or mild.
- Treatment: Treatment involves antibiotics, such as ciprofloxacin or doxycycline, and may require surgical debridement of the affected area. The prognosis is generally good with appropriate treatment, but untreated cases can lead to systemic spread and potentially fatal outcomes.
💛 (e) Immunoglobulin G
Immunoglobulin G (IgG)*:
👉1. Function: IgG is the most abundant class of antibodies in the bloodstream, constituting approximately 75% of the total immunoglobulin pool.
- Structure: It is composed of two identical heavy chains and two identical light chains, forming a Y-shaped molecule.
- Functions: IgG plays a crucial role in the adaptive immune response by:
- Neutralizing toxins and viruses.
- Facilitating phagocytosis by binding to pathogens and marking them for destruction by immune cells.
- Activating the complement system to enhance pathogen clearance.
- Long-term Immunity: IgG provides long-term immunity against pathogens and can cross the placenta, conferring passive immunity to the fetus during pregnancy.
- Subtypes: IgG has four subclasses (IgG1, IgG2, IgG3, IgG4), each with unique effector functions and distribution in the body.
- Diagnostic Significance: Measurement of IgG levels in blood serum is important for diagnosing various infectious diseases, autoimmune disorders, and immune deficiencies.
💛 (f) Immunity
Immunity*:
👉
1. *Definition*: Immunity refers to the body’s ability to resist and defend against infectious agents, toxins, and other foreign substances that can cause harm.
2. *Types of Immunity*:
– *Innate Immunity*: Present from birth, innate immunity provides immediate, nonspecific defense mechanisms against a wide range of pathogens. It includes physical barriers (e.g., skin, mucous membranes), cellular components (e.g., macrophages, natural killer cells), and soluble factors (e.g., complement proteins, cytokines).
– *Adaptive Immunity*: Also known as acquired immunity, adaptive immunity develops throughout life in response to exposure to specific pathogens or antigens. It involves highly specialized immune cells (e.g., B cells, T cells) that can recognize and target specific antigens, leading to a tailored immune response.
3. *Components of Adaptive Immunity*:
– *Humoral Immunity*: Mediated by antibodies produced by B cells, humoral immunity targets pathogens or toxins circulating in the bloodstream or body fluids.
– *Cellular Immunity*: Mediated by T cells, cellular immunity targets infected or abnormal cells by directly killing them or secreting cytokines to activate other immune cells.
4. *Memory and Specificity*: Adaptive immunity exhibits memory and specificity, meaning that upon re-exposure to a previously encountered pathogen or antigen, the immune system can mount a faster and more robust response tailored to that specific threat.